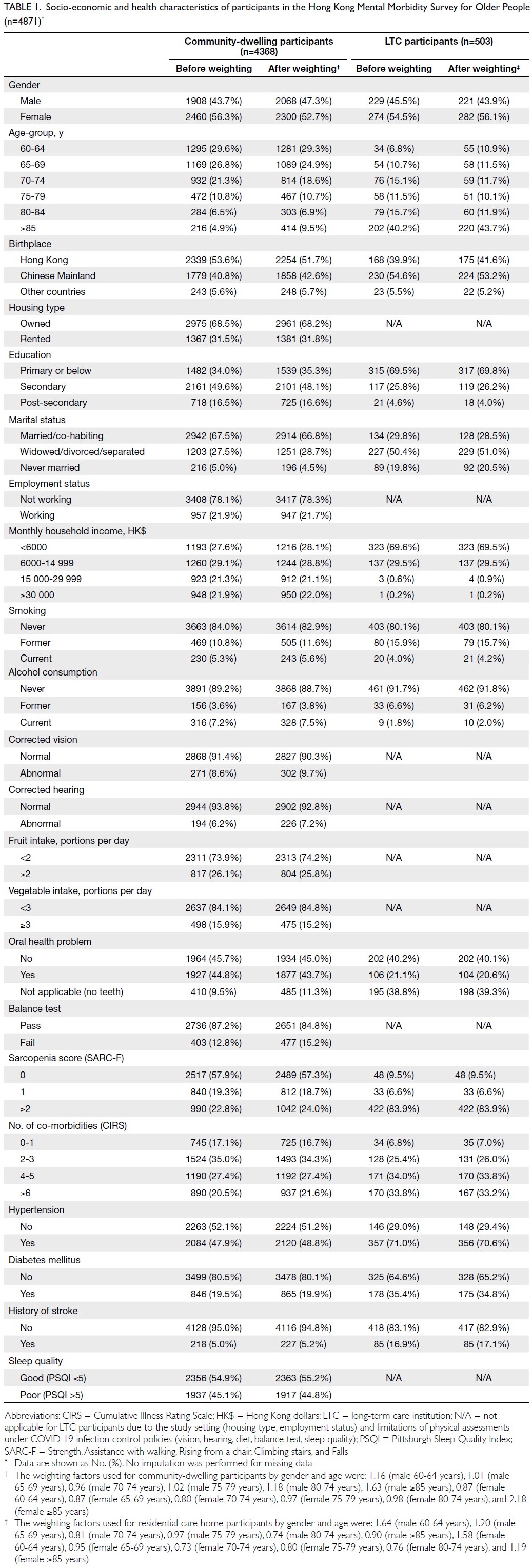
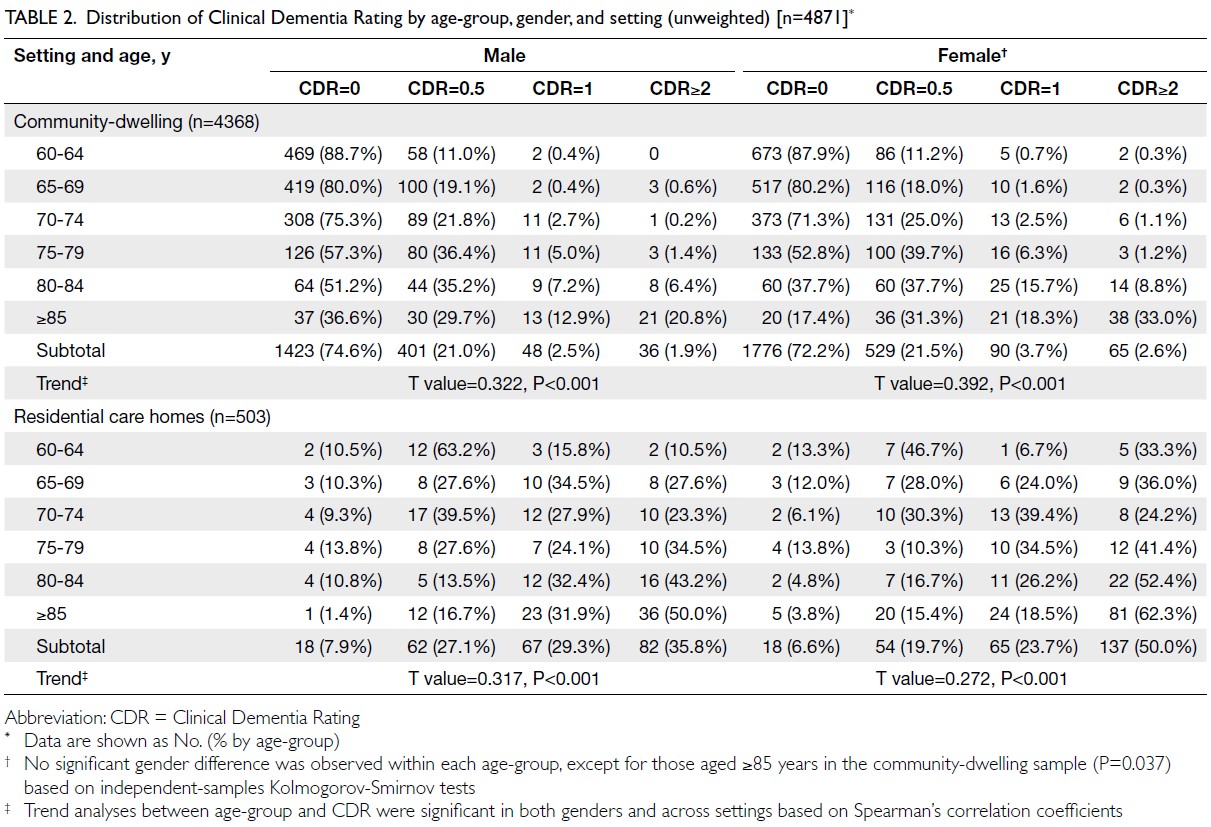
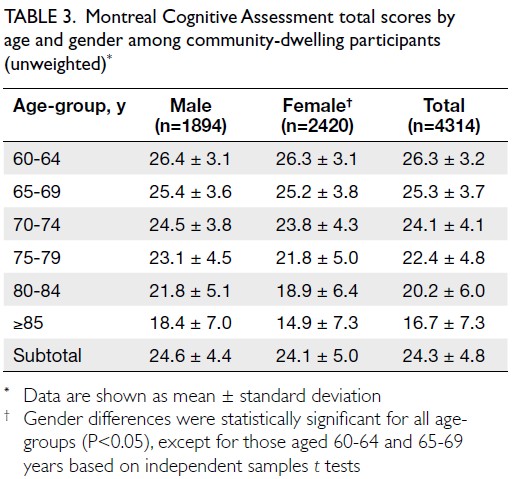
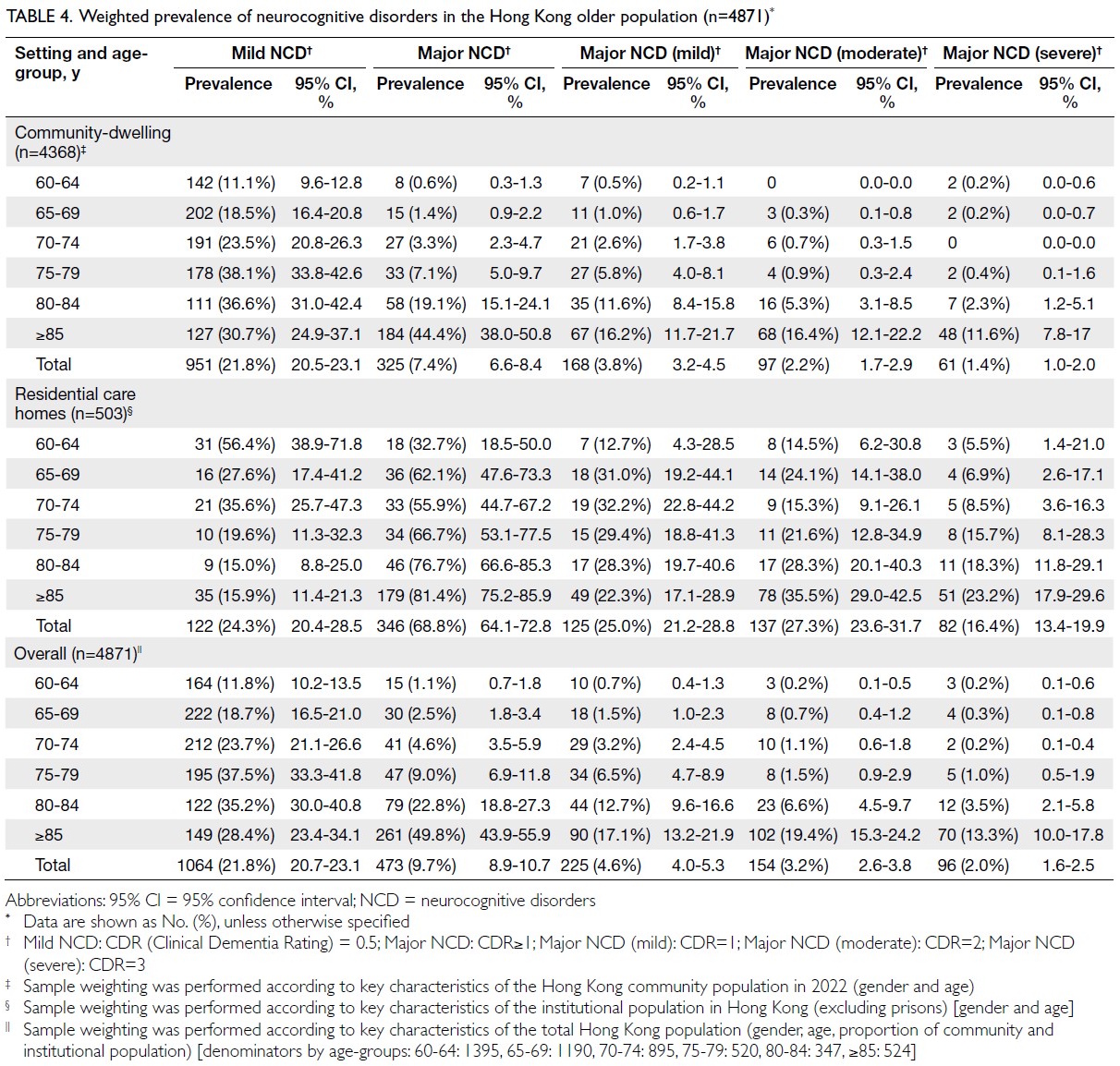
Obesity-driven thyroid cancer burden in middle-aged and older populations: temporal trends and projected trajectories based on the Global Burden of Disease study
Hong Kong Med J 2026 Apr;32(2):135–43 | Epub 16 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Obesity-driven thyroid cancer burden in middle-aged
and older populations: temporal trends and projected trajectories based on the Global Burden of Disease study
Bo Jiang, BMed1 #; Jing Li, MMed2 #; Xi Sun, PhD3 #; Jingyu Qu, BMed4 #; Jing Li, BMed5; Li Li, BMed6; Dong Cai, BMed6; Yanli Zhao, MCM7; Jia Tian, PhD8; Jie Lian, BMed9; Xuhua Liu, BMed10; Chunhuo Zhang, MSc11; Shuying Niu, BSc12; Ying Yu, BMed13; Jun Han, PhD14
1 Department of Clinical Medicine, The Fourth Affiliated Hospital, Harbin
Medical University, Harbin, China
2 Department of Endocrinology and Metabolism, Heilongjiang Academy of
Traditional Chinese Medicine, Harbin, China
3 Department of Research, The Fourth Affiliated Hospital, Harbin Medical
University, Harbin, China
4 Clinical Medicine, Harbin Medical University, Harbin, China
5 Xinlin District People’s Hospital, Xinlin, China
6 Department of Geriatrics, Xinlin District People’s Hospital, Xinlin, China
7 Department of General Practice, Dawusu Town Health Center, Xinlin, China
8 Department of Nephrology, The Fourth Affiliated Hospital, Harbin Medical University, Harbin, China
9 Department of Ultrasound, The Fourth Affiliated Hospital, Harbin Medical University, Harbin, China
10 Department of Geriatrics, The Fourth Affiliated Hospital, Harbin Medical University, Harbin, China
11 Da Hinggan Ling Health Commission, Jagdaqi, China
12 Xinlin Health Commission, Xinlin, China
13 Department of Ophthalmology, The Fourth Affiliated Hospital, Harbin Medical University, Harbin, China
14 Department of Endocrinology and Metabolism, The Fourth Affiliated Hospital, Harbin Medical University, Harbin, China
# Equal contribution
Corresponding authors: Prof Jun Han (hanjun198887@sina.com); Prof Ying Yu (happyhatty@163.com)
Abstract
Introduction: High body mass index (BMI) in
middle-aged and older individuals (≥40 years) is
a leading risk factor for thyroid cancer–related
morbidity and mortality; however, the quantifiable
impact of elevated BMI on disability-adjusted life
years (DALYs) and mortality in ageing populations
remains underexplored. This study comprehensively
evaluated the global burden of thyroid cancer
attributable to elevated BMI by integrating past
epidemiological trends, demographic variability
and risk attribution models, and provided relevant
projected trajectories using data from the Global
Burden of Disease (GBD) study.
Methods: We analysed mortality, DALYs, years
of life lost (YLLs) and years lived with disability
(YLDs). Temporal trends in disease burden from
1990 to 2021 were examined using linear regression
models. Cluster analysis was used to assess region-specific
burdens across GBD study regions. Finally,
projections of future disease burden from 2022 to
2050 were generated using autoregressive integrated
moving average and exponential smoothing models.
Results: In 2021, high BMI contributed to 5255
thyroid cancer–related deaths (age-standardised
mortality rate: 0.06 per 100 000) and 144 955 DALYs
(age-standardised rate: 1.68 per 100 000); women
and low-middle Socio-demographic Index regions
were identified as high-risk subgroups. Projections
indicate continued increases in mortality and overall
disease burden through 2050.
Conclusion: Substantial geographical heterogeneity
in thyroid cancer burden was observed across
GBD regions. Interventions targeting high-risk
demographic groups and regions should be
prioritised to reduce this growing disease burden.
New knowledge added by this study
- This is the first study to confirm the quantifiable impact of elevated body mass index (BMI) on disability-adjusted life years and mortality in ageing demographic groups.
- This study comprehensively evaluated the global disease burden of thyroid cancer attributable to elevated BMI by integrating epidemiological trends, demographic variability, and risk attribution models from 1990 to 2021.
- The Hong Kong Government could propose sex- and age-specific prevention strategies, metabolic risk mitigation, and early detection protocols to address the increasing public health threat posed by obesity-driven thyroid cancer.
- The Hong Kong Government could prioritise interventions in high-risk demographic groups and regions to reduce this growing disease burden.
Introduction
Thyroid cancer is one of the most common endocrine
malignancies,1 and its global incidence has steadily
increased in recent decades.2 This rise is primarily
attributed to an increased incidence of papillary
thyroid carcinoma.3 Patients with papillary thyroid
carcinoma generally have a favourable prognosis;
with appropriate treatment, the 5-year survival
rate exceeds 98.3%.4 Most known or suspected risk
factors for thyroid cancer, such as age, sex, race or
ethnicity, and family history, are non-modifiable.5
However, changes in other factors, including
obesity, cancer detection, iodine intake and ionising
radiation, may influence the observed incidence,
mortality and disability-adjusted life years (DALYs)
of thyroid cancer over time. It is well documented
that elevated body mass index (BMI) influences
cancer development across multiple malignancies.6 7
We speculate that these aggregated trends do
not accurately reflect the true disease burden in
populations with high BMI, particularly among
middle-aged and older adults, because existing
studies address only the heterogeneity of thyroid
cancer incidence across regions.8 9 To address this
gap, we used data from the Global Burden of Disease
(GBD) 2021 study to systematically analyse the
burden of thyroid cancer among middle-aged and older adults with high BMI from 1990 to 2021, and
to project the future burden from 2022 through
2050. This analysis will assist policymakers in
assessing thyroid cancer burden, evaluating the
progress in targeted therapies, allocating resources,
and formulating evidence-based policies.
Methods
Overview
The GBD 2021 study conducted a comprehensive
assessment of health loss across 204 countries and
territories, encompassing 369 diseases, injuries and
impairments, as well as 88 risk factors, using updated
epidemiological data and refined standardisation
methodologies.10 The GBD database employs
sophisticated methods to address missing data and
adjust for confounding factors. Detailed descriptions
of the GBD study design and analytical approaches
have been extensively documented.10 Data used
in the present study were obtained from the GBD
2021 database (https://ghdx.healthdata.org/gbd-2021), which contains no personally identifiable information.
Socio-demographic Index
The Socio-demographic Index (SDI) quantifies
regional development status using aggregated
measures of fertility rate, per capita income and
educational attainment, scaled from 0 (least
developed) to 1 (most developed). Within the GBD
2021 framework, countries were classified into five
SDI tiers: high (>0.81), high-middle (0.70-0.81),
middle (0.61-0.69), low-middle (0.46-0.60), and low
(<0.46).10
Time series analysis
A time series comprises systematically recorded
data points indexed at uniform temporal intervals
(daily, monthly or yearly), enabling the identification
of temporal patterns and trends. To forecast
thyroid cancer burden metrics, we implemented
autoregressive integrated moving average (ARIMA)
models, which incorporate systematic evaluation
of autoregressive, moving average and differencing
parameters to optimise predictive accuracy.11
Study data
In this study, the burden of thyroid cancer associated
with high BMI among populations aged <40 years was
assumed to be negligible. Consequently, individuals
aged ≥40 years were stratified into 12 age-groups.
Statistical analyses
The statistical analysis evaluated global deaths,
DALYs, years of life lost (YLLs), years lived with
disability (YLDs), and age-standardised rates for high-BMI–related thyroid cancer in middle-aged
and older populations (2021), stratified by age, sex,
SDI, region and country. Temporal trends (1990-2021) were analysed globally and across subgroups
using linear regression models to estimate annual
percentage changes.12 Decomposition analysis using
the Das Gupta method (modified by Cheng et al
[2020])13 14 isolated the effects of population ageing,
population growth and epidemiological changes
on variations in disease burden. The ARIMA and
exponential smoothing models were used to project
future disease burden (2022-2050). All analyses
were performed using R software (version 4.0.2) for
database management, computation and validation.
Results
Disease burden of thyroid cancer attributable
to high body mass index in middle-aged and older populations
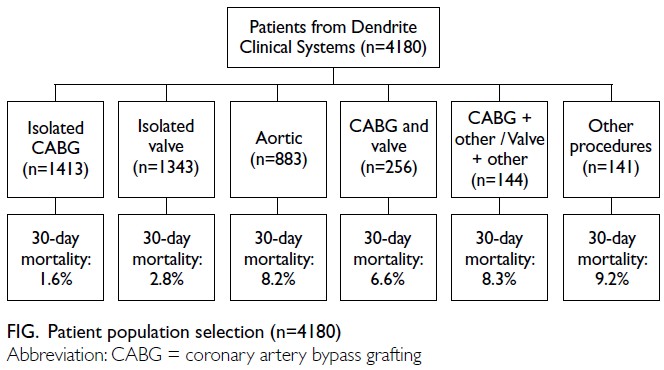
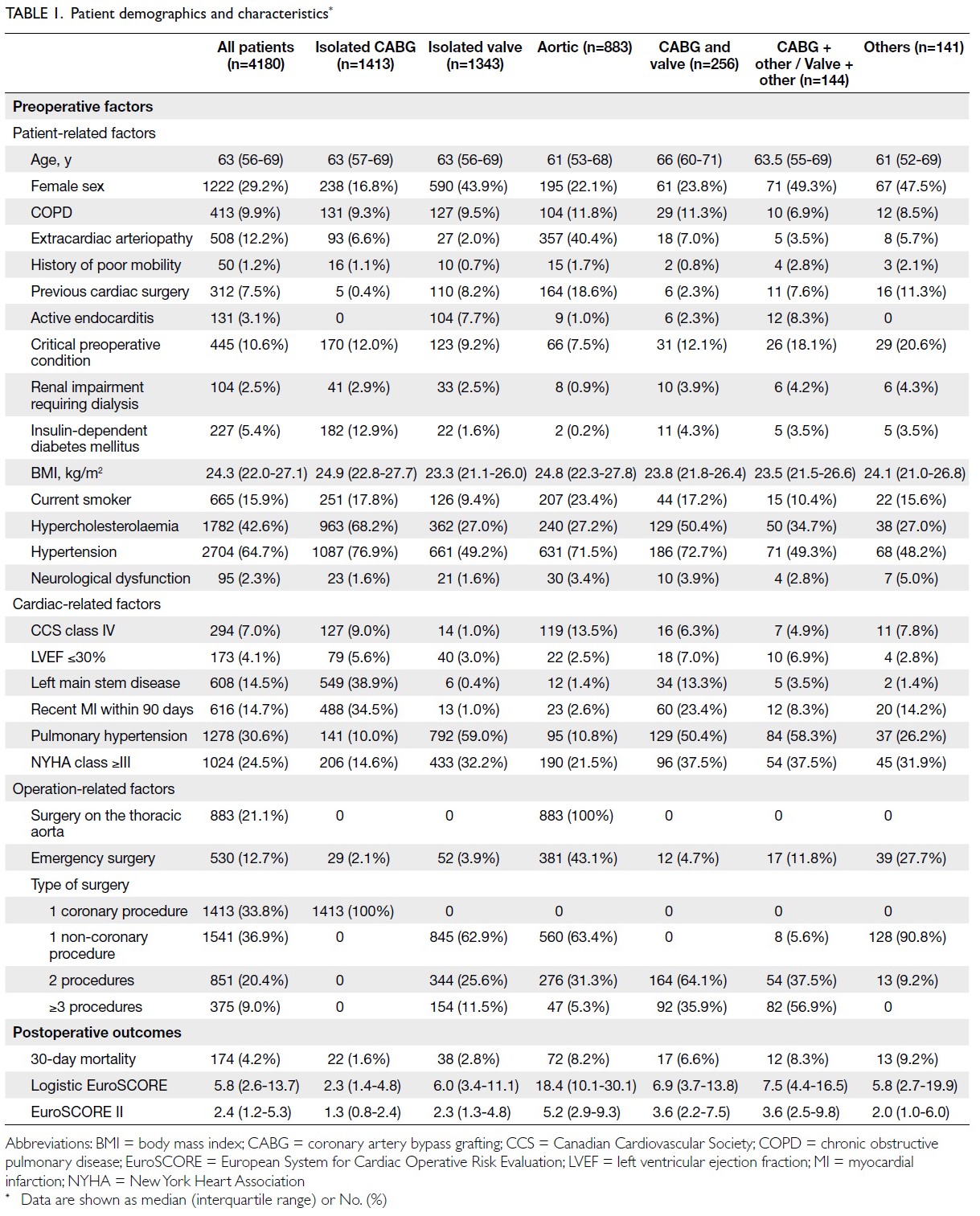
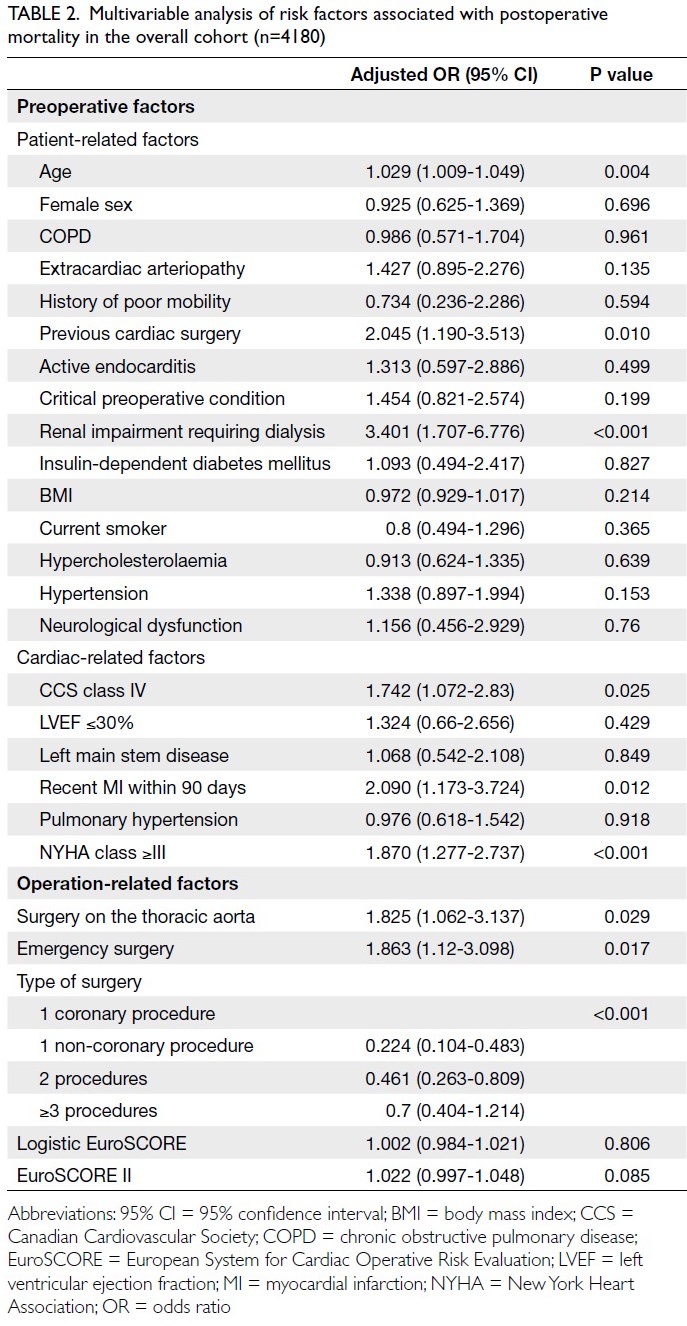
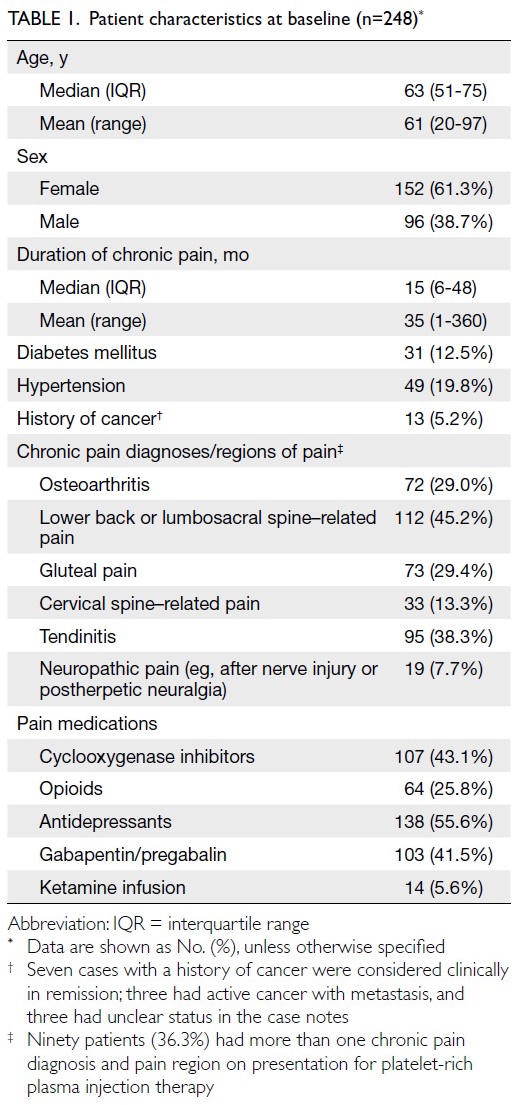
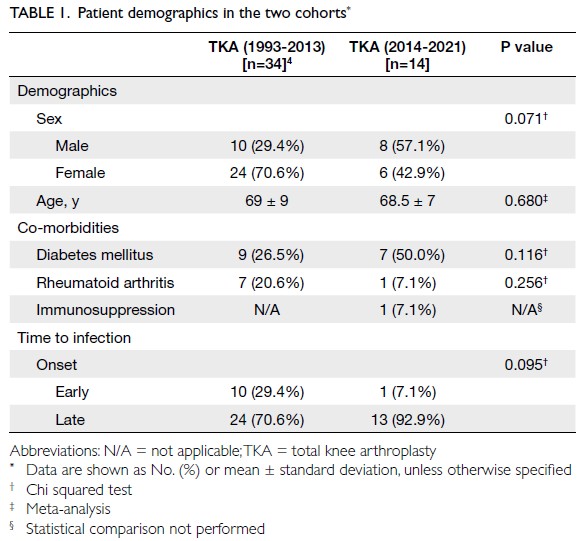
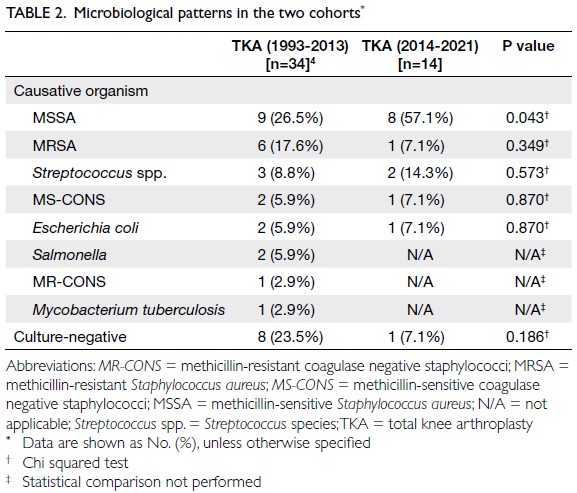
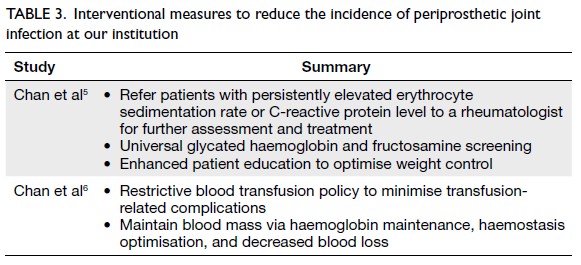
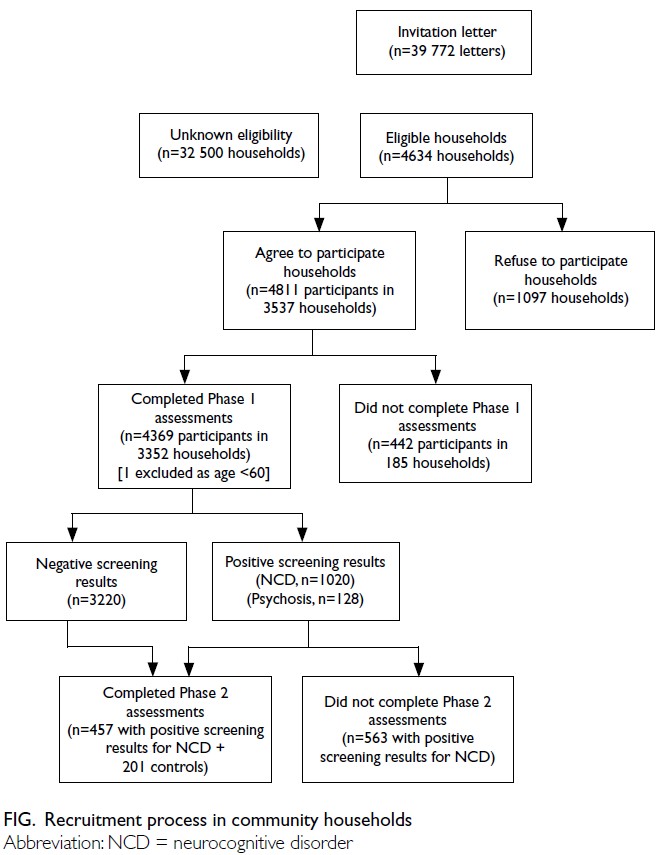
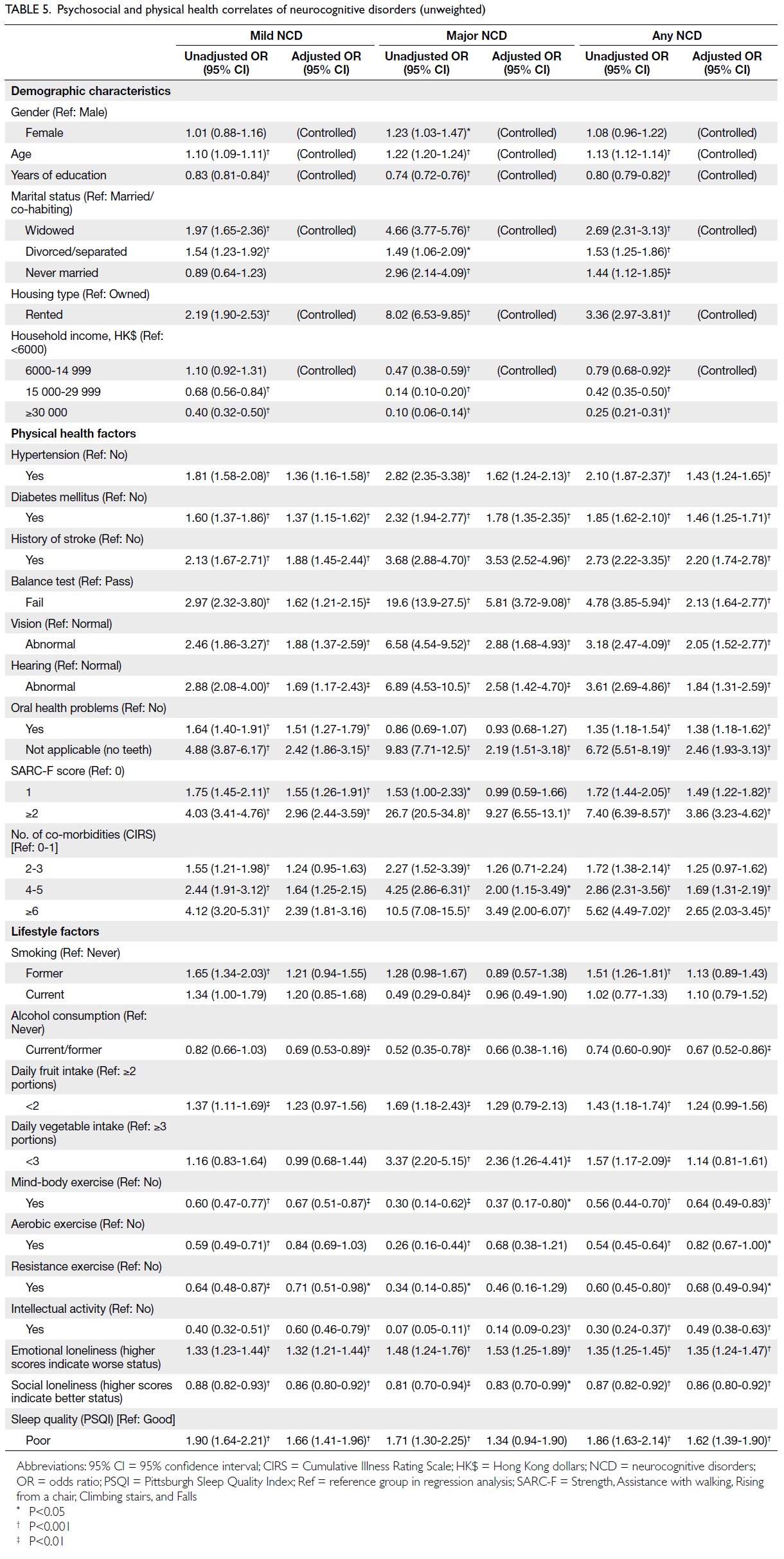
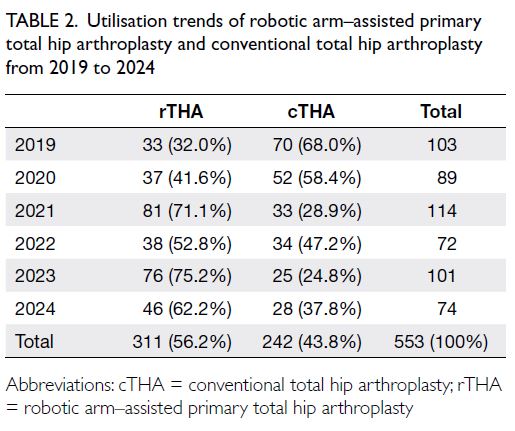
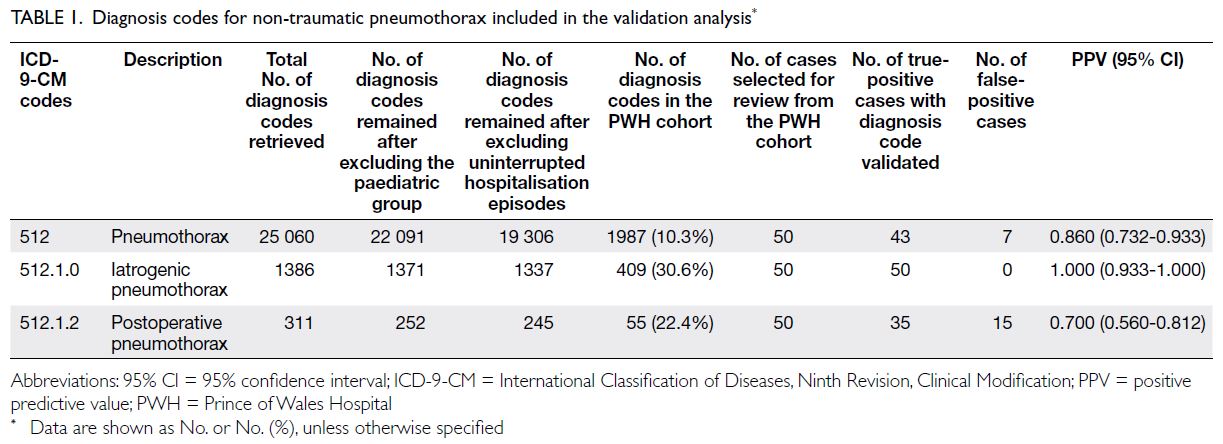
Globally, high-BMI–associated thyroid cancer among middle-aged and older populations caused 5255 deaths (95% uncertainty interval [95% UI]=3914-6653), with an age-standardised mortality rate of
0.06 per 100 000 (95% UI=0.05-0.08) [Table 1]. The
number of attributable DALYs totalled 144 955
(95% UI=109 230-184 747), corresponding to an
age-standardised DALY rate of 1.68 per 100 000
(95% UI=1.26-2.14) [Table 2]. Specifically, the number
of YLDs reached 15 968 (95% UI=10 370-23 793; age-standardised
rate: 0.18 per 100 000 [95% UI=0.12-0.28]) [Table 3], whereas YLLs constituted 128 986
(95% UI=96 149-162 365; age-standardised rate: 1.50
per 100 000 [95% UI=1.12-1.88]) [Table 4]. Age-standardised
mortality, DALY, and YLL rates for
high-BMI–related thyroid cancer increased with
age, whereas YLD rates peaked in the 70-74 years
age-group before declining. Non-linear age-specific
patterns were observed for absolute case counts:
deaths and DALYs peaked in the 55-59 years age-group
(Table 2), YLDs in the 55-59 years age-group
(Table 3), and YLLs in the 65-69 years age-group (Table 4). In 2021, female predominance was evident
across all metrics. Females accounted for 61.37%
of deaths, 60.79% of DALYs, 66.23% of YLDs, and
60.12% of YLLs. Geographically, middle-SDI regions
had the highest absolute burden (1649 deaths; 47 448
DALYs), whereas high-middle SDI regions exhibited
the highest age-standardised mortality (0.06 per
100 000) and DALY rates (1.63 per 100 000) [Tables 1 and 2].
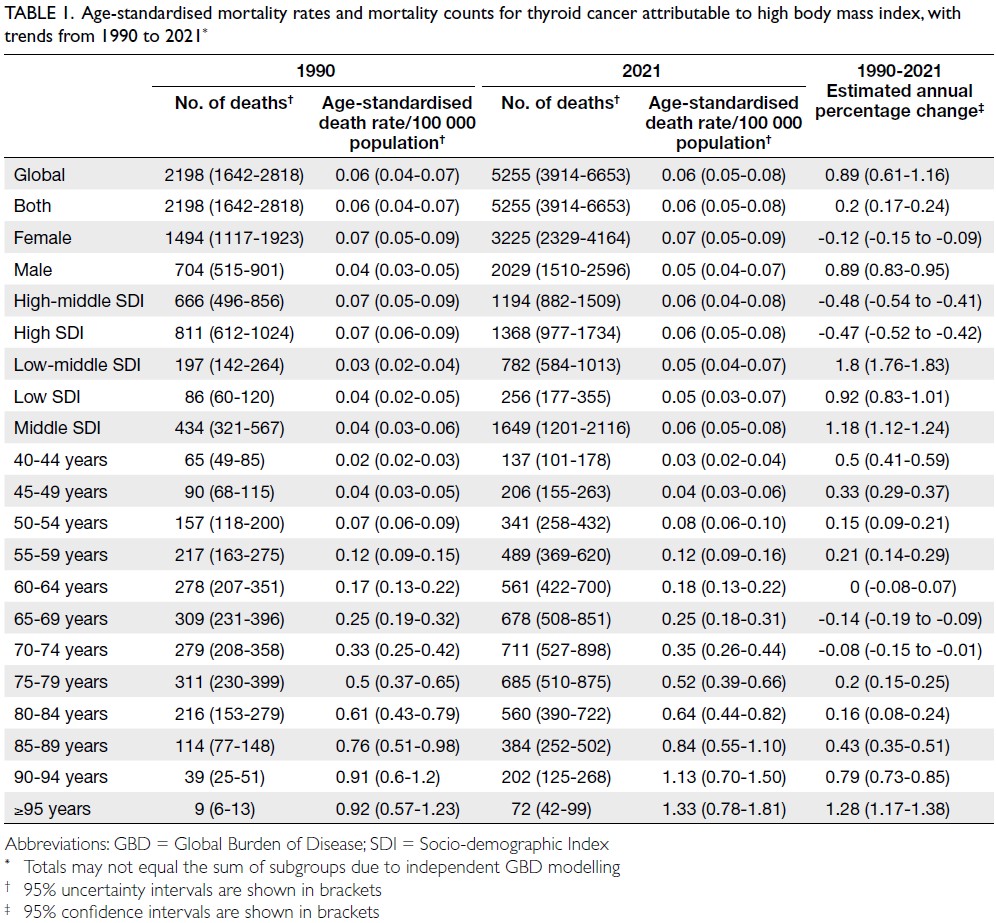
Table 1. Age-standardised mortality rates and mortality counts for thyroid cancer attributable to high body mass index, with trends from 1990 to 2021
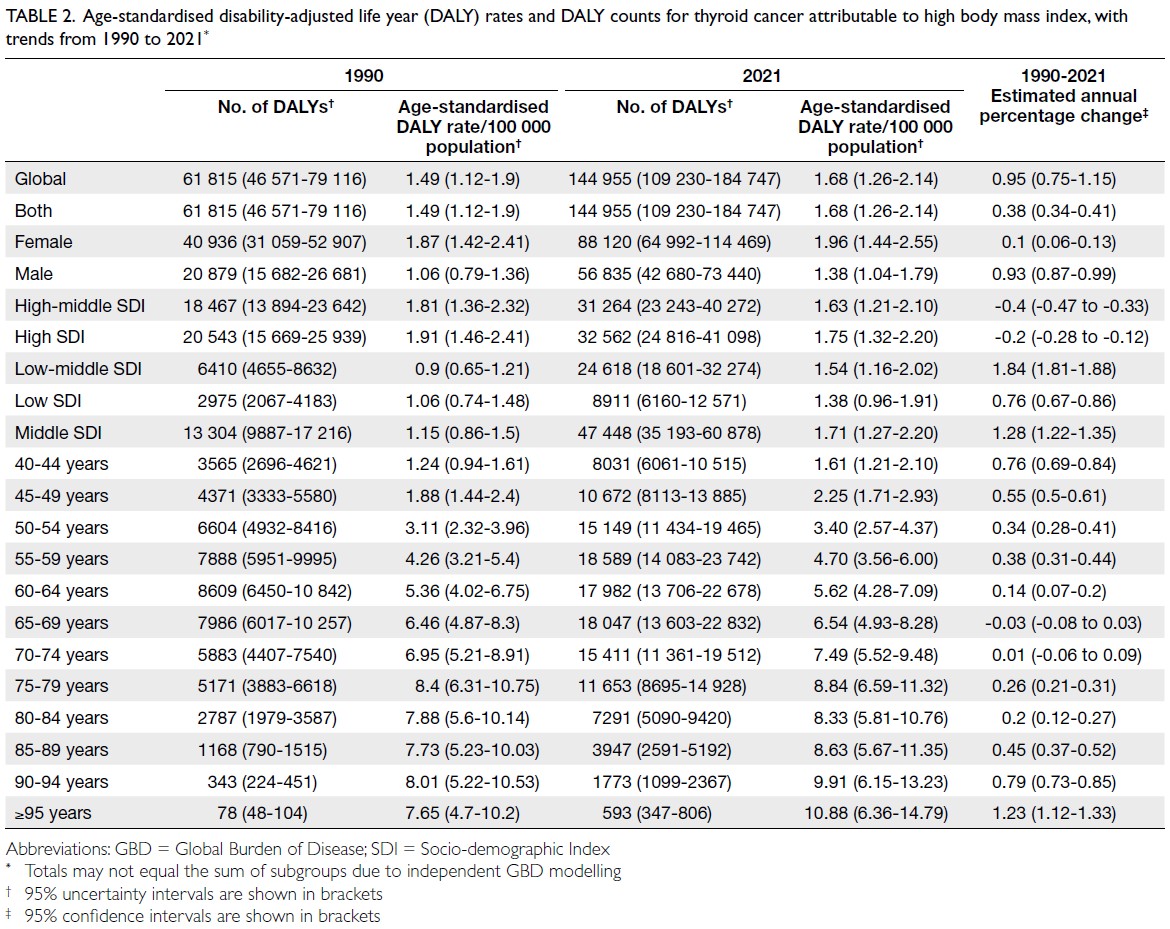
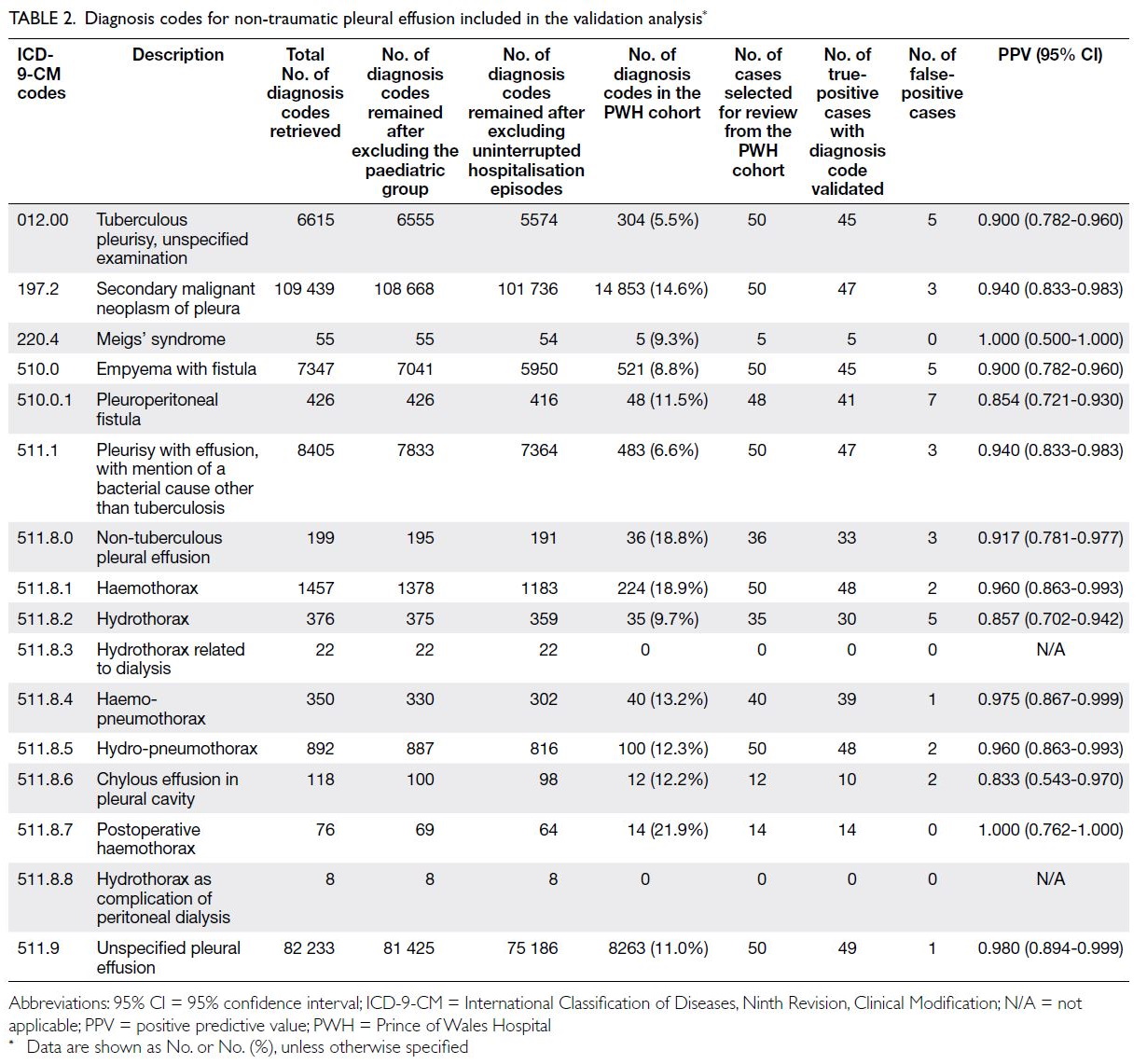
Table 2. Age-standardised disability-adjusted life year (DALY) rates and DALY counts for thyroid cancer attributable to high body mass index, with trends from 1990 to 2021
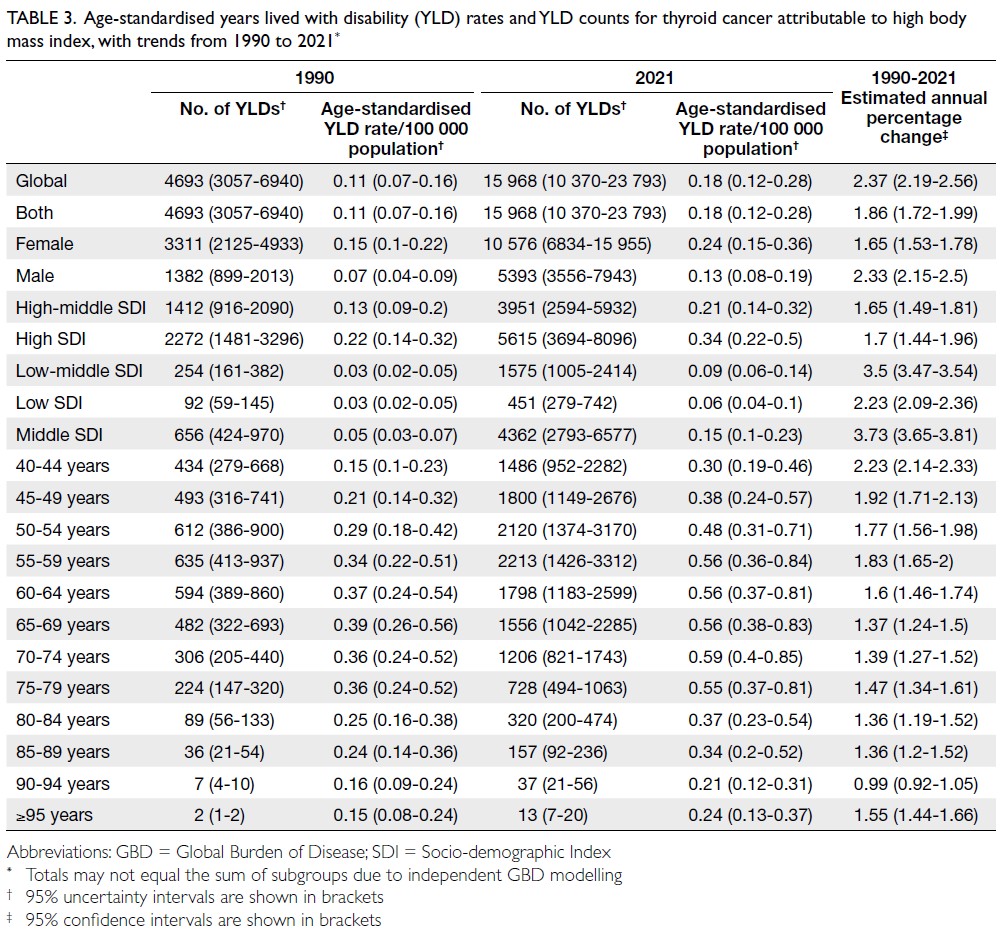
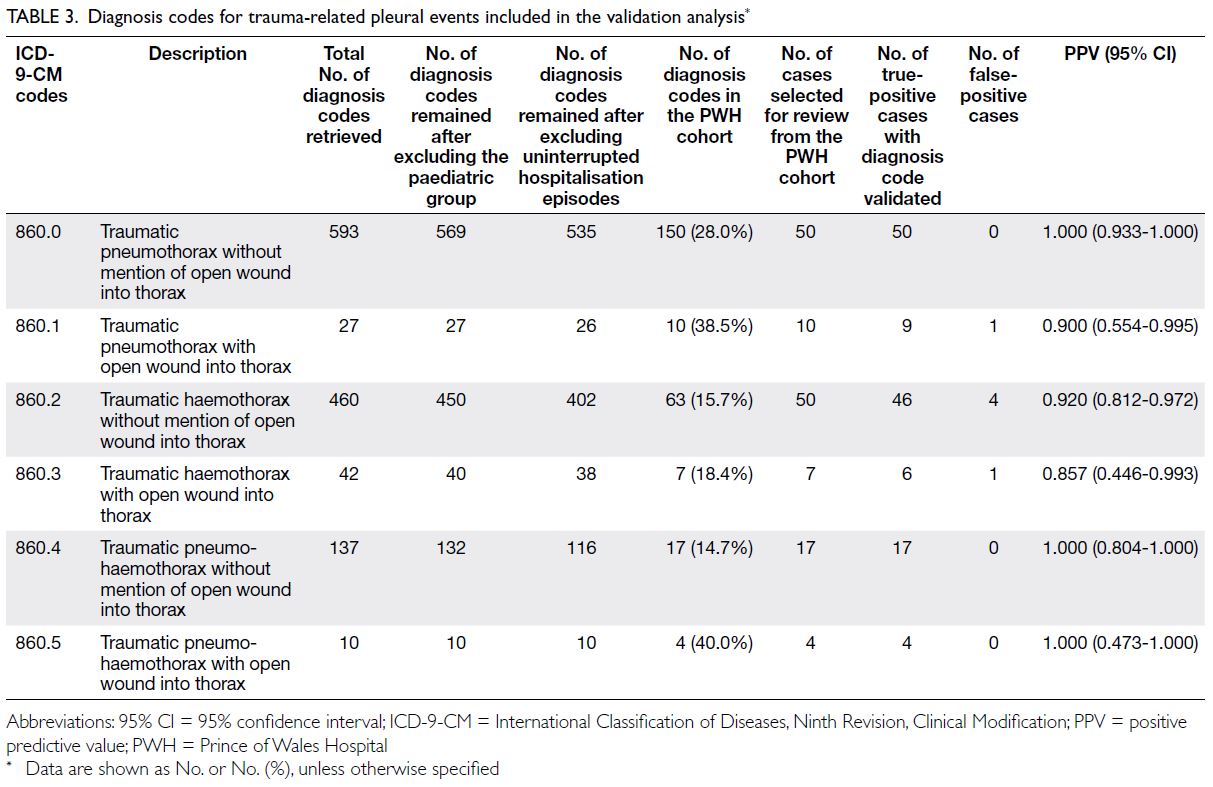
Table 3. Age-standardised years lived with disability (YLD) rates and YLD counts for thyroid cancer attributable to high body mass index, with trends from 1990 to 2021
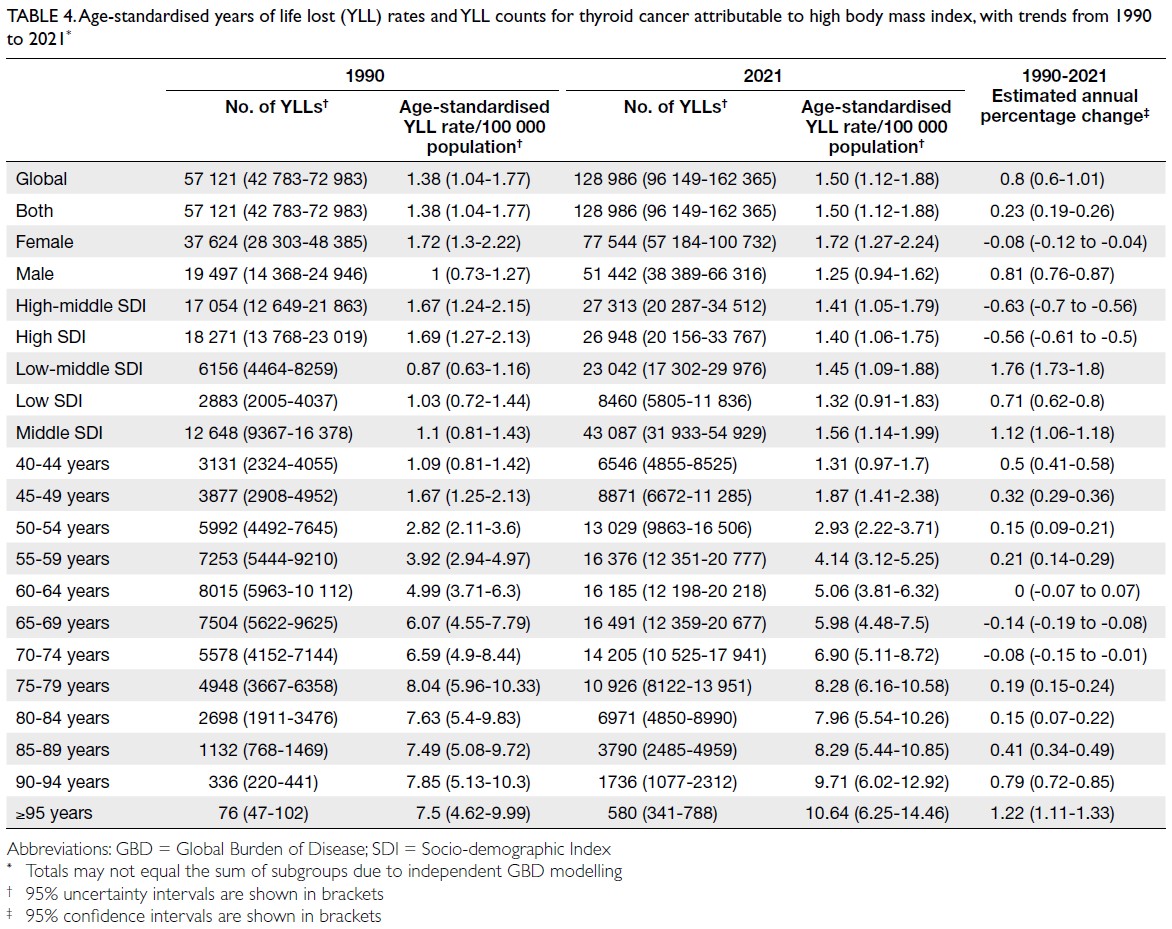
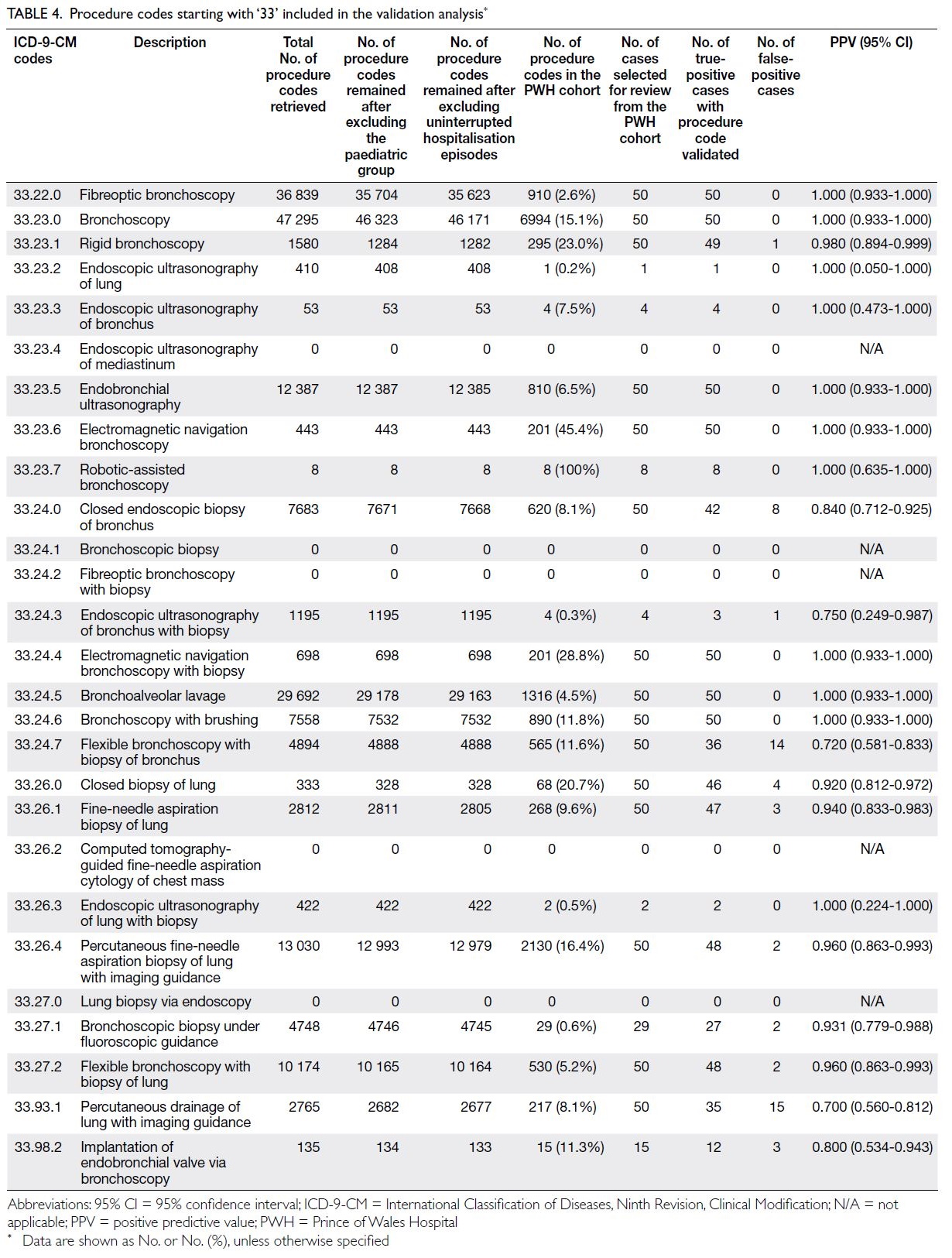
Table 4. Age-standardised years of life lost (YLL) rates and YLL counts for thyroid cancer attributable to high body mass index, with trends from 1990 to 2021
Globally, substantial disparities in the burden
of high-BMI–related thyroid cancer were observed
across 50 GBD regions in 2021. Asia displayed
the highest absolute burden, with 75 130 DALYs
(95% UI=54 305-97 695), 2601 deaths (95% UI=1884-3397), 7596 YLDs (95% UI=4771-11 735), and
67 533 YLLs (95% UI=48 806-88 249), whereas
Oceania reported the lowest values, with 163 DALYs
(95% UI=102-238) and five deaths (95% UI=3-7).
Age-standardised rates revealed regional heterogeneity: Andean Latin America exhibited
among the highest age-standardised rates for DALYs
(4.26 per 100 000; 95% UI=3.03-5.89), deaths (0.16
per 100 000; 95% UI=0.12-0.23) and YLLs (3.98 per
100 000; 95% UI=2.80-5.48).
At the national level, China recorded
the highest number of DALYs (23 684;
95% UI=16 056-32 507) and deaths (871; 95% UI=588-1177), followed by India (11 546-20 676 DALYs; 506
deaths). Fiji (Oceania) demonstrated the highest
age-standardised DALY rate (6.07 per 100 000;
95% UI=3.76-8.98), exceeding that of Ecuador
(South American, Andean region: 5.12 per 100 000;
95% UI=3.57-6.92). China also exhibited the
highest global YLD (2871; 95% UI=1780-4650)
and YLL (20 814; 95% UI=13 923-28 116) counts,
reflecting its disproportionate burden among
ageing populations with elevated BMI (online supplementary Fig 1).
Temporal trends in disease burden
attributable to high body mass index–related
thyroid cancer in middle-aged and older
populations
From 1990 to 2021, the numbers of thyroid cancer–related deaths, DALYs, YLDs, and YLLs increased
worldwide, reflecting a growing public health burden.
Age-standardised rates for all metrics showed an
overall increasing trend during this period, indicating
persistent elevations in mortality and morbidity risk
independent of population ageing. These findings
suggest that the increasing disease burden cannot be
attributed solely to demographic expansion, but may
involve synergistic drivers such as environmental
exposures or lifestyle changes (online supplementary Fig 2).
Sex-specific disparities were evident in temporal
progression patterns; men displayed concurrent
upward trends in age-standardised morbidity and mortality rates, as well as case numbers, highlighting
sex-dimorphic epidemiological mechanisms (online supplementary Fig 3).
Age-stratified analysis revealed differential
temporal patterns: middle-aged cohorts (40-44
years) showed relatively stable age-standardised
rates in later decades despite increasing case
counts, suggesting improved early detection or risk
mitigation. Conversely, older populations (70-79
years) experienced concurrent increases in age-standardised
morbidity metrics and absolute case
counts, indicating that disease progression may be
driven by ageing-related physiological vulnerabilities
and prolonged exposure to risk factors (online supplementary Fig 4).
Geographical heterogeneity was observed
across SDI regions. High- and high-middle-SDI
regions achieved declining age-standardised rates
despite increasing case numbers, likely reflecting advances in healthcare infrastructure and diagnostic
precision. In contrast, low-middle- and low-SDI
regions experienced parallel increases in age-standardised
rates and absolute case counts,
underscoring the compounding effects of limited
healthcare access, delayed diagnosis, and unmitigated
metabolic risk factors (online supplementary Fig 5).
Globally, thyroid cancer–related DALYs,
deaths, YLDs, and YLLs among middle-aged and
older populations with elevated BMI increased
from 1990 to 2021. Population growth was the
predominant driver of these increases, followed by
epidemiological changes and population ageing.
High- and high-middle-SDI areas were
primarily influenced by population growth and
epidemiological shifts, with minimal contribution
from ageing. Middle-SDI regions showed substantial
contributions from all three factors—population
growth, epidemiological changes, and ageing. In low-middle- and low-SDI regions, population
growth remained the dominant driver, although
epidemiological changes and ageing also contributed
(online supplementary Fig 6). Sex-specific
decomposition revealed differing contribution
patterns. Among women, population growth was
the primary driver of the burden, with additional
contributions from epidemiological changes and
smaller effects from ageing. In contrast, men
exhibited a dual-driver pattern in which population
growth and epidemiological changes jointly
accounted for most of the burden, while ageing
played a lesser role (online supplementary Fig 7).
Predicted results for 2022 to 2050
The ARIMA model projections for 2022 to 2050
indicated that the numbers of deaths, DALYs,
YLDs, and YLLs related to thyroid cancer are
expected to increase in both sexes. Corresponding age-standardised rates demonstrated relative
stabilisation in women and an upward trend in men;
these patterns were corroborated by exponential
smoothing models (online supplementary Fig 8).
Discussion
Thyroid cancer is one of the most prevalent endocrine
malignancies worldwide. Although the overall
survival rate remains relatively high, its increasing
incidence in many countries and regions, particularly
in more developed nations, has become a growing
public health concern.15 Globally, approximately
560 000 new cases of thyroid cancer are diagnosed
annually, with a female-to-male incidence ratio of
around 3:1.16 17 18 Concurrently, obesity has emerged
as a major clinical and public health challenge,
exhibiting rapid growth trends in both developed and
developing countries. The impact of elevated BMI
on cancer development has been well documented
across multiple malignancies.7 However, the specific
mechanisms underlying the association between
elevated BMI and thyroid carcinogenesis remain
poorly understood, constituting a critical knowledge
gap that warrants further investigation.
According to the Global Burden of Disease
Study 2021, thyroid cancer incidence rates have
shown a sustained annual increase worldwide, with
particularly pronounced rises among women in
countries such as the United States and South Korea.19
This trend has been attributed to advances in early
screening and diagnostic technologies. Furthermore,
active surveillance has been recommended for the
management of papillary microcarcinoma; these
minimally invasive tumours frequently demonstrate
favourable prognoses and indolent biological
progression. This strategy effectively avoids
overtreatment while reducing unnecessary surgical
and therapeutic interventions.20 In recent years,
China has updated its clinical guidelines for thyroid
nodule management, emphasising early screening
protocols, standardisation of fine-needle aspiration
biopsy, and personalised treatment planning.21 These
revised strategies, particularly in the management of
differentiated thyroid cancer, have further improved
patient survival and quality of life.
The present study leveraged the GBD 2021 study database to evaluate thyroid cancer–related
mortality, DALYs, YLDs, and YLLs among middle-aged
and older individuals (≥40 years) with elevated
BMI from 1990 to 2021. The results revealed an
age-dependent increase in mortality burden, with
DALYs and YLDs peaking in the 55-59-year age-group
and YLLs reaching maximal levels in the 65-69-year age-group. Notably, populations aged ≥85
years displayed attenuated disease burden metrics
in absolute counts, potentially reflecting diminished
physiological reserves that mask the clinical manifestations of malignancy, thereby contributing
to diagnostic delays, therapeutic limitations, and
exacerbated mortality. These findings underscore
the critical interplay between ageing, metabolic
risk, and healthcare accessibility in shaping thyroid
cancer outcomes among high-BMI populations.
This study confirmed persistent sex disparities
in thyroid cancer burden, with the incidence and
prevalence among women consistently exceeding
those among men across all regions in both 1990 and
2021. These disparities likely arise from an interplay
of biological and socio-cultural mechanisms. Central
to this imbalance are hormonal drivers—particularly
oestrogen fluctuations during the menopausal
transition—which may promote thyroid cell
proliferation and oncogenesis. In addition to these
biological factors, sex-specific lifestyle patterns,
such as chronic stress, dietary habits, and exposure
to environmental pollutants, may further increase
tumourigenic risk. Underlying both dimensions,
socio-cultural determinants affecting healthcare
access may introduce diagnostic ascertainment
bias, potentially obscuring the true epidemiological
landscape.17
Notably, our analysis revealed a progressive rise
in the proportions of obesity-driven thyroid cancer
mortality and DALY proportions from 1990 to 2021;
men exhibited a substantially greater escalation in
burden relative to women.22 These patterns align
with global epidemiological shifts—47.1% and
27.5% increases in adult and childhood obesity
prevalence, respectively, from 1980 to 2013.23 Such
trends likely contribute to the disproportionate
increase in thyroid cancer burden among male
populations. Mechanistically, prolonged obesity
may synergise with age-related endocrine alterations
through amplified metabolic dysregulation and
chronic inflammation, thereby promoting thyroid
carcinogenesis in ageing men.24
Low-SDI regions display lower overall thyroid
cancer incidence rates but significantly faster growth
than high-SDI regions. In contrast, high-SDI regions
show stable or marginally declining incidence trends,
potentially attributable to advanced healthcare
infrastructure and higher health literacy, which
enable early diagnosis and optimised management.
These disparities emphasise the critical role of socio-economic
development in shaping the epidemiology
of thyroid cancer. Prioritising SDI-stratified
interventions tailored to regional healthcare capacity
and risk profiles could enhance the precision and
impact of burden-mitigation strategies.25
Projections indicate escalating thyroid cancer
mortality, DALYs, YLLs, and YLDs from 2022 to 2050,
with progressive increases among men but stable
rates among women, consistent with documented
epidemiological trajectories.9 This rising burden in
middle-aged and older populations with elevated BMI likely reflects synergistic interactions involving
demographic ageing, the proliferation of high-risk
behaviours, and socio-economic transitions. These
forecasts highlight the urgent need to integrate
tertiary prevention strategies with early-stage
interventions targeting metabolic risk mitigation
and diagnostic optimisation.
This investigation is strengthened by its
rigorous analysis of the obesity-driven thyroid
cancer burden in ageing populations using the
GBD 2021 study dataset (1990-2021), coupled with
comprehensive male patient data to delineate sex-specific
epidemiological trajectories. However, the
findings are tempered by several methodological
constraints. The lack of histopathological subtype
classification, such as papillary, follicular, or
anaplastic variants, limits prognostic granularity.
Additionally, there was limited consideration of
modifiable risk factors, including gradients of
radiation exposure and fluctuations in dietary
iodine intake, which may synergistically interact
with metabolic risk. Furthermore, this study did
not fully disentangle how therapeutic advances (eg,
surgical techniques, radiotherapy protocols, and
molecular-targeted agents) modulate longitudinal
disease burden. Collectively, these gaps underscore
the imperative for intervention-focused studies
integrating molecular stratification and context-specific
risk profiling to refine clinical management
paradigms.
Obesity is associated with an increased risk of
at least 13 cancers (eg, endometrial, oesophageal,
renal, and pancreatic adenocarcinomas;
hepatocellular carcinoma; gastric cancer; colorectal
cancer; postmenopausal breast cancer; ovarian
cancer; gallbladder cancer; and thyroid cancer).
Its biological mechanisms are multifactorial,
mainly involving chronic inflammation, hormonal
dysregulation, and metabolic disturbances: (1)
long-term systemic inflammation may impair tissue
repair capacity and promote tumour development26;
(2) disruption of hormonal balance, as adipose tissue
is a major source of aromatase activity that converts
androgens to oestrogen, thereby increasing the risk
of hormone-related malignancies27; and (3) increased
visceral and subcutaneous fat accumulation may
promote metabolic abnormalities that contribute
to the development of liver, endometrial, and other
cancers.28
Therefore, from a public health perspective,
efforts should be strengthened to increase
awareness of the association between obesity and
cancer, promote health education, and encourage
population-level weight control to reduce cancer
incidence. From an individual perspective, effective
weight management should be emphasised,
including reducing the intake of high-fat and high-sugar
foods, adopting a high-fibre, low-calorie diet, increasing physical activity, and undergoing regular
health screening (including monitoring body weight,
waist circumference, blood glucose, lipid levels,
and liver and kidney function) to reduce the risk of
obesity-related tumours.
Conclusion
Significant geographical heterogeneity in thyroid
cancer burden was observed across GBD regions.
These findings underscore the urgent need for sex- and
age-specific prevention strategies, metabolic risk
mitigation, and early detection protocols to address
the growing public health threat posed by obesity-driven
thyroid cancer. Interventions targeting high-risk
demographic groups and regions should be
prioritised to reduce this increasing disease burden.
Author contributions
Concept or design: B Jiang, S Niu, X Sun, J Qu, Y Yu, C Zhang, J Li2, J Han.
Acquisition of data: J Li5, L Li, D Cai, Y Zhao, J Tian, J Lian, X Liu.
Analysis or interpretation of data: All authors.
Drafting of the manuscript: B Jiang, S Niu, X Sun, J Qu.
Critical revision of the manuscript for important intellectual content: Y Yu, C Zhang, J Li2, J Han.
Acquisition of data: J Li5, L Li, D Cai, Y Zhao, J Tian, J Lian, X Liu.
Analysis or interpretation of data: All authors.
Drafting of the manuscript: B Jiang, S Niu, X Sun, J Qu.
Critical revision of the manuscript for important intellectual content: Y Yu, C Zhang, J Li2, J Han.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Funding/support
This research was funded by the Heilongjiang Province
Postdoctoral Research Start-up Fund (Ref No.: 21042240063),
the Fundamental Research Funds for the Provincial
Universities (Ref No.: 2023-KYYWF-0236), the Fundamental
Research Funds for the Provincial Universities (Ref No.:
2023-KYYWF-0234), and the Excellent Youth Program of
the Fourth Affiliated Hospital of Harbin Medical University,
China (Ref No.: HYDSYYXQN2023015). The funders had no role in study design, data collection, analysis, interpretation, or manuscript preparation.
Ethics approval
Detailed descriptions of the Global Burden of Disease (GBD)
study design and analytical approaches have been extensively
documented in existing GBD publications. The data used
in this study were obtained from the GBD 2021 study database (https://ghdx.healthdata.org/gbd-2021), which contains no personally identifiable information. All original studies were
reviewed and approved by the relevant ethics committees.
Supplementary material
The supplementary material was provided by the authors, and
some information may not have been peer reviewed. Accepted
supplementary material will be published as submitted by the
authors, without any editing or formatting. Any opinions
or recommendations discussed are solely those of the author(s) and are not endorsed by the Hong Kong Academy
of Medicine and the Hong Kong Medical Association.
The Hong Kong Academy of Medicine and the Hong Kong
Medical Association disclaim all liability and responsibility
arising from any reliance placed on the content.
References
1. Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA 2024;331:425-35. Crossref
2. Pizzato M, Li M, Vignat J, et al. The epidemiological landscape of thyroid cancer worldwide: GLOBOCAN estimates for incidence and mortality rates in 2020. Lancet Diabetes Endocrinol 2022;10:264-72. Crossref
3. Sipos JA, Mazzaferri EL. Thyroid cancer epidemiology and prognostic variables. Clin Oncol (R Coll Radiol) 2010;22:395-404. Crossref
4. Nabhan F, Dedhia PH, Ringel MD. Thyroid cancer, recent advances in diagnosis and therapy. Int J Cancer 2021;149:984-92. Crossref
5. Kitahara CM, Sosa JA. The changing incidence of thyroid cancer. Nat Rev Endocrinol 2016;12:646-53. Crossref
6. Li C, Zhang J, Dionigi G, Liang N, Guan H, Sun H. Uncovering the connection between obesity and thyroid cancer: the therapeutic potential of adiponectin receptor agonist in the AdipoR2-ULK axis. Cell Death Dis 2024;15:708. Crossref
7. Lengyel E, Makowski L, DiGiovanni J, Kolonin MG. Cancer as a matter of fat: the crosstalk between adipose tissue and tumors. Trends Cancer 2018;4:374-84. Crossref
8. Deng Y, Li H, Wang M, et al. Global burden of thyroid cancer from 1990 to 2017. JAMA Netw Open 2020;3:e208759. Crossref
9. Zhai M, Zhang D, Long J, et al. The global burden of thyroid cancer and its attributable risk factor in 195 countries and territories: a systematic analysis for the Global Burden of Disease study. Cancer Med 2021;10:4542-54. Crossref
10. GBD 2021 Diseases and Injuries Collaborators. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease study 2021. Lancet 2024;403:2133-61. Crossref
11. Lou HR, Wang X, Gao Y, Zeng Q. Comparison of ARIMA model, DNN model and LSTM model in predicting disease burden of occupational pneumoconiosis in Tianjin, China. BMC Public Health 2022;22:2167. Crossref
12. Wang F, Ma B, Ma Q, Liu X. Global, regional, and national burden of inguinal, femoral, and abdominal hernias: a systematic analysis of prevalence, incidence, deaths, and DALYs with projections to 2030. Int J Surg 2024;110:1951-67. Crossref
13. Cheng X, Yang Y, Schwebel DC, et al. Population ageing and mortality during 1990-2017: a global decomposition analysis. PLoS Med 2020;17:e1003138. Crossref
14. Das Gupta P. Standardization and decomposition of rates from cross-classified data. Genus 1994;50:171-96.
15. Chen DW, Lang BH, McLeod DS, Newbold K, Haymart MR. Thyroid cancer. Lancet 2023;401:1531-44. Crossref
16. Suteau V, Munier M, Briet C, Rodien P. Sex bias in differentiated thyroid cancer. Int J Mol Sci 2021;22:12992. Crossref
17. Shobab L, Burman KD, Wartofsky L. Sex differences in differentiated thyroid cancer. Thyroid 2022;32:224-35. Crossref
18. Remer LF, Lee CI, Picado O, Lew JI. Sex differences in papillary thyroid cancer. J Surg Res 2022;271:163-70. Crossref
19. Murray CJ; GBD 2021 Collaborators. Findings from the Global Burden of Disease study 2021. Lancet 2024;403:2259-62. Crossref
20. Vaccarella S, Franceschi S, Bray F, Wild CP, Plummer M, Dal Maso L. Worldwide thyroid-cancer epidemic? The increasing impact of overdiagnosis. N Engl J Med 2016;375:614-7. Crossref
21. Wang H, Wang J, Wang X, et al. Comments on National guidelines for diagnosis and treatment of thyroid cancer 2022 in China (English version). Chin J Cancer Res 2022;34:447-50. Crossref
22. Chong B, Jayabaskaran J, Kong G, et al. Trends and predictions of malnutrition and obesity in 204 countries and territories: an analysis of the Global Burden of Disease study 2019. EClinicalMedicine 2023;57:101850. Crossref
23. Ng M, Fleming T, Robinson M, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease study 2013. Lancet 2014;384:766-81. Crossref
24. Schmid D, Ricci C, Behrens G, Leitzmann MF. Adiposity and risk of thyroid cancer: a systematic review and meta-analysis. Obes Rev 2015;16:1042-54. Crossref
25. Zhou T, Wang X, Zhang J, et al. Global burden of thyroid cancer from 1990 to 2021: a systematic analysis from the Global Burden of Disease study 2021. J Hematol Oncol 2024;17:74. Crossref
26. Iyengar NM, Gucalp A, Dannenberg AJ, Hudis CA. Obesity and cancer mechanisms: tumor microenvironment and inflammation. J Clin Oncol 2016;34:4270-6. Crossref
27. Engin A. Obesity-associated breast cancer: analysis of risk factors and current clinical evaluation. Adv Exp Med Biol 2024;1460:767-819. Crossref
28. Sohn W, Lee HW, Lee S, et al. Obesity and the risk of primary liver cancer: a systematic review and meta-analysis. Clin Mol Hepatol 2021;27:157-74. Crossref