Hong Kong Med J 2016 Jun;22(3):223–30 | Epub 6 May 2016
DOI: 10.12809/hkmj154730
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE CME
Effect of non-invasive prenatal testing as a
contingent approach on the indications for
invasive prenatal diagnosis and prenatal
detection rate of Down’s syndrome
KO Kou, MRCOG, FHKAM (Obstetrics and Gynaecology)1;
CF Poon, MSc Health Care (Nursing), RDMS (OB/GYN & FE)1;
SL Kwok, RNM, Master of Nursing (Clinical Leadership)1;
Kelvin YK Chan, BSc, PhD2;
Mary HY Tang, FRCOG, FHKAM (Obstetrics and Gynaecology)2;
Anita SY Kan, MRCOG, FHKAM (Obstetrics and Gynaecology)2;
KY Leung, MD, FHKAM (Obstetrics and Gynaecology)1
1 Department of Obstetrics and Gynaecology, Queen Elizabeth Hospital,
Jordan, Hong Kong
2 Prenatal Diagnostic Laboratory, Tsan Yuk Hospital and Department of
Obstetrics and Gynaecology, Queen Mary Hospital, Hong Kong
Corresponding author: Dr KY Leung (leungky1@ha.org.hk)
Abstract
Introduction: In Hong Kong, universal combined
first-trimester screening for Down’s syndrome was
started as a ‘free service’ in July 2010. Non-invasive
prenatal testing was available as a self-financed
item in August 2011. This study aimed to determine
whether the introduction of non-invasive prenatal
testing as a contingent approach influenced the
indications for invasive prenatal diagnosis and the
consequent prenatal detection of Down’s syndrome.
Methods: This historical cohort study was
conducted at the Prenatal Diagnosis Clinic of Queen
Elizabeth Hospital in Hong Kong. We compared
the indications for invasive prenatal diagnosis and
prenatal detection of Down’s syndrome in singleton
pregnancies 1 year before and 2 years following
the availability of non-invasive prenatal testing as
a contingent test after a positive aneuploidy test.
All pregnant women who attended our hospital
for counselling about universal Down’s syndrome
screening between August 2010 and July 2013 were
recruited.
Results: A total of 16 098 women were counselled.
After the introduction of non-invasive prenatal
testing, the invasive prenatal diagnosis rate for a
positive aneuploidy screening reduced from 77.7%
in 2010-11 to 68.8% in 2012-13. The new combined
conventional plus non-invasive prenatal testing
strategy was associated with a lower false-positive
rate (6.9% in 2010-11 vs 5.2% in 2011-12 and 4.9%
in 2012-13). There was no significant increase in
invasive prenatal diagnosis for structural anomalies
over the years. There was no significant trend
in the overall prenatal detection rate of Down’s
syndrome (100% 1 year before vs 89.1% 2 years after
introduction of non-invasive prenatal testing). Four
(2.6%) of 156 women who underwent non-invasive
prenatal testing for a screen-positive result had a
high-risk result for trisomy 21, which was subsequently
confirmed by invasive prenatal diagnosis. There
were no false-negative cases.
Conclusion: The introduction of non-invasive
prenatal testing as a contingent approach reduced
the invasive prenatal diagnosis rate for a positive
aneuploidy screening without affecting the invasive
prenatal diagnosis rate for structural anomalies or
the overall detection rate of fetal Down’s syndrome.
New knowledge added by this study
- Introduction of non-invasive prenatal testing (NIPT) decreased overall invasive prenatal diagnosis (IPD) rate for a positive aneuploidy screening without affecting the IPD rate for structural anomalies.
- NIPT as a contingent approach does not affect the overall detection rate of fetal Down’s syndrome.
- NIPT provides a safe contingent approach for a positive aneuploidy screening. This is particularly relevant in centres with a high false-positive rate following conventional screening for Down’s syndrome.
- Extending the indications of NIPT from high-risk to intermediate- or low-risk women with a view to increasing the prenatal detection rate of Down’s syndrome requires further evaluation.
Introduction
Over the last 30 years, there has been a shift in
clinical practice away from performing an invasive
prenatal diagnosis (IPD) on the basis of maternal
age to a non-invasive screening method1 2 3 4 5 with
improving performance.6 7 8 9 10 11 12 Initially, the introduction
of second-trimester screening (STS) resulted in an
overall increase in the number of IPD tests, mainly
amniocentesis, for women aged <35 years with
screen-positive result.1 The later implementation of
combined first-trimester screening (cFTS) caused
a mild and gradual decrease in the number of IPD
and amniocentesis, but an increase in the number or
proportion of chorionic villus sampling (CVS) tests.1 2 3
The recent introduction of non-invasive prenatal
testing (NIPT) has resulted in a rapid decrease in the
number of invasive tests including amniocentesis
and CVS within a short period of time.5 13 14 Such
non-invasive testing has a higher sensitivity (95.5-100% vs 85-90%) and a lower false-positive rate
(0.002-0.2% vs 3-5%) than traditional non-invasive
screening methods for Down’s syndrome,6 7 8 9 10 11 12 and is
well accepted by women5 and physicians.15
Although not as a free service, NIPT has been
available in Hong Kong since August 2011. In our
previously published study,16 we showed that the
introduction of NIPT as a contingent test resulted
in a significant decrease in IPD by 16.3% and
25.6% in the first and second year, respectively. It
remained unclear, however, whether this change in
practice affected the overall prenatal detection rate
of Down’s syndrome and whether the indication for
IPD because of a scan abnormality was increased. In
the present study, using the same study population
as before, we aimed to review all the indications for
IPD and to determine any alteration in the overall
prenatal detection of Down’s syndrome before and
after the introduction of NIPT as a contingent
approach.
Methods
This historical cohort study was conducted at the Prenatal Diagnosis Clinic of Queen Elizabeth Hospital, Hong Kong. All pregnant women who attended our hospital for counselling on prenatal testing for Down’s syndrome between August 2010 and July 2013 were recruited. The utilisation of conventional screening, NIPT, and IPD for Down’s syndrome and
other aneuploidies in all singleton pregnancies were
included. Our hospital is one of the largest referral
public hospitals in Hong Kong with around 6000
deliveries a year. This study was approved by the
Research Ethics Committee of Kowloon Central/Kowloon East Cluster, Hong Kong. Informed consent
was not required for this retrospective study.
Since 1 July 2010, universal prenatal screening
for Down’s syndrome with cFTS between 11 weeks
and 13 weeks and 6 days or STS has been offered to all pregnant women
after adequate counselling. Combined first-trimester
screening includes fetal nuchal translucency (NT)
measurement, and free beta–human chorionic
gonadotrophin and pregnancy-associated
plasma protein-A assessment. The
gestational age is determined by an ultrasound
examination (crown rump length in the first or head
biometry in the second trimester) shortly after the
first antenatal visit.
All NT measurements were performed by
trained midwives and doctors who were Fetal
Medicine Foundation–certified or accredited
as maternal fetal medicine (MFM) subspecialists.
In our hospital, MFM team doctors counselled
screen-positive (risk ≥1 in 250 in cFTS) women
about different options including IPD with CVS
or amniocentesis, or no further prenatal invasive
testing. After August 2011, the option of self-financed
NIPT was also discussed at the request of
patients.
Most commercial NIPT was based on massively
parallel sequencing with ‘shotgun’ counting of
all cell-free DNA sequences while others involve
‘targeted’ counting of specific DNA sequences. A
usual NIPT report includes the risk for trisomies 21,
18, and 13. Sex chromosomal or other abnormalities
are also reported if identified on NIPT. If NIPT
demonstrated a high risk for trisomy 21, 18 or 13,
confirmatory IPD was required. After undergoing
NIPT in the private sector, the woman would be
followed up, counselled by doctors, rescanned for
any structural fetal anomaly and offered an option
of invasive testing, regardless of the results of NIPT,
at no charge. We advised women with fetal NT ≥3.5
mm or structural abnormalities to undergo IPD
rather than NIPT given their higher risk of atypical
chromosome abnormalities that might not be
picked up by the latter.17 In addition, IPD would be
offered (a) if first-trimester or routine mid-trimester
anomaly scan showed an abnormality, (b) for genetic
diseases like thalassaemia, (c) if there was a positive
family history, or (d) rarely, if there was maternal anxiety after
adequate counselling.
Chromosome analysis was mainly performed
by the prenatal diagnostic laboratory of Tsan Yuk
Hospital and a small proportion at Prince of Wales
Hospital, Hong Kong. These two laboratories are
accredited by professional bodies in providing
prenatal diagnostic tests and serve the local obstetric
units. Analyses included G-banding chromosome
analyses and quantitative fluorescence polymerase
chain reaction for rapid aneuploidy detection. All
pregnancy outcomes were traced by reviewing
hospital records or phone contact in women who
delivered outside this hospital.
We determined the number of fetuses and
newborns with Down’s syndrome prenatally or
postnatally, the rate of different prenatal tests for
Down’s syndrome, and the number of IPD that
were needed to diagnose one fetus with Down’s
syndrome. We also reviewed the indications for
IPD and classified them as one of the following
priorities: high risk for trisomy 21, 18, 13 or other
aneuploidy on NIPT, increased NT (≥3.5 mm),
structural anomalies on ultrasound, parental carrier
of or previous pregnancies with abnormal karyotype,
positive aneuploidy screening, maternal age ≥35
years, or others. If NT was increased and cFTS
trisomy 21 risk was high, increased NT would be
selected as the sole indication for IPD.
Statistical analyses
With the use of descriptive statistics and Chi squared
test for linear trend, the rates of conventional
screening, NIPT, IPD, and prenatally diagnosed
Down’s syndrome were compared 1 year before
and 2 years after NIPT introduction. All statistical analyses were performed using the Statistical Package for the
Social Sciences (Windows version 21.0; SPSS Inc,
Chicago [IL], US).
Results
A total of 16 098 women with singleton pregnancies
were counselled on prenatal testing for Down’s
syndrome. Although the total proportion of women
who underwent screening remained around 97%
from 2010 to 2013, cFTS increased from 84.4% in
2010-11 to 90.5% in 2012-13 and STS decreased
from 12.1% to 7.1% in the same period (P<0.001)
[Table 1]. With a sharp increase in the use of NIPT
in the screen-positive group (0%, 0.8%, and 1.9% in
2010-11, 2011-12, 2012-13, respectively; P<0.001),
the rate of IPD and CVS dropped from 7.6% and
4.2% in 2010-11 to 6.0% and 2.5% in 2012-13,
respectively while the amniocentesis rate fluctuated.
The actual number of IPD remained similar over the
years though the rate declined, probably because
of an increasing number of screenings from 4288 in
2010-11 to 5618 in 2012-13 (Table 1).
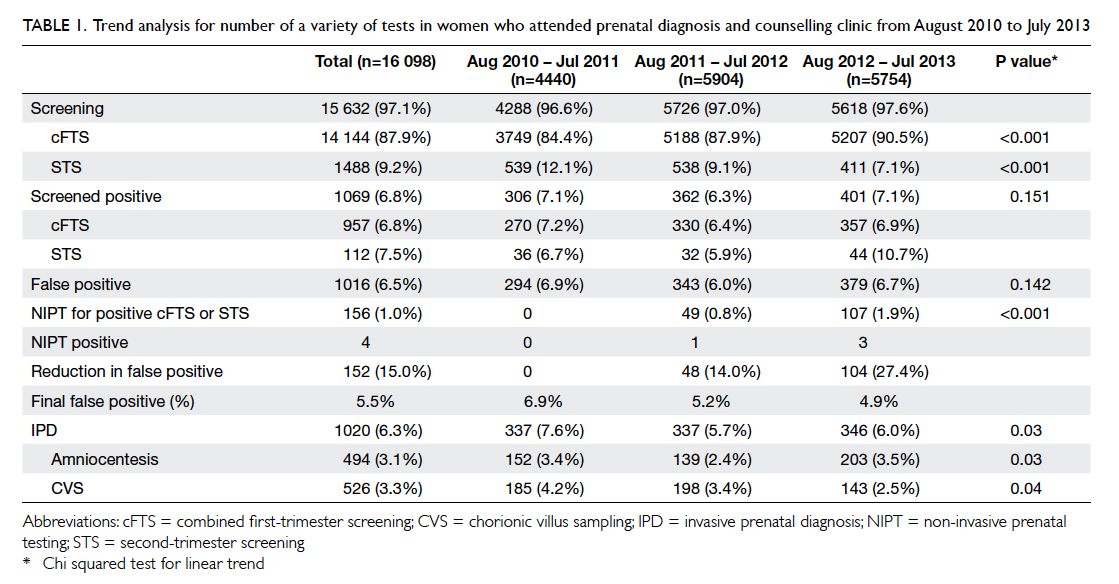
Table 1. Trend analysis for number of a variety of tests in women who attended prenatal diagnosis and counselling clinic from August 2010 to July 2013
Indications for invasive prenatal diagnosis
There was a significant decrease in IPD for positive
aneuploidy screening from 77.7% in 2010-11 to
68.8% in 2012-13 (P=0.005). There was no significant
increase in IPD for structural anomalies over the
years (Table 2).
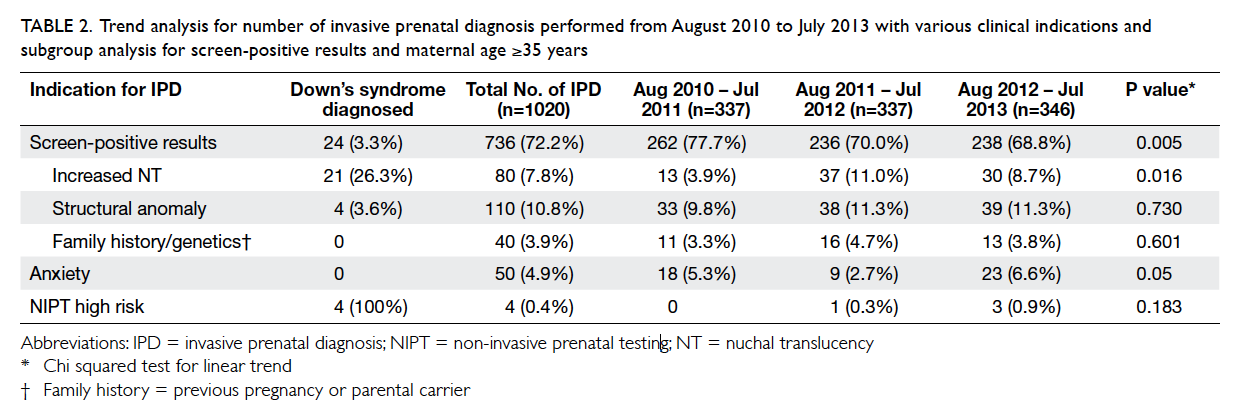
Table 2. Trend analysis for number of invasive prenatal diagnosis performed from August 2010 to July 2013 with various clinical indications and subgroup analysis for screen-positive results and maternal age ≥35 years
Prenatal detection of Down’s syndrome
There was no significant trend in the overall prenatal
diagnosis/detection rate of Down’s syndrome
before and after the availability of NIPT (Table 3).
The number of IPD required to diagnose one case
of Down’s syndrome decreased from 28 to 16 over
the same period of time, though the trend was not
significant, probably because of the small sample
size (Table 3). There were nine newborn infants
with Down’s syndrome over the 3 years. In two
cases, women declined IPD despite a positive Down’s
syndrome screening result. In another two cases that
were included in the 45 diagnosed prenatally,
women chose to continue their pregnancy after IPD
of Down’s syndrome. The remaining five cases
were screened negative and diagnosed postnatally (Fig).
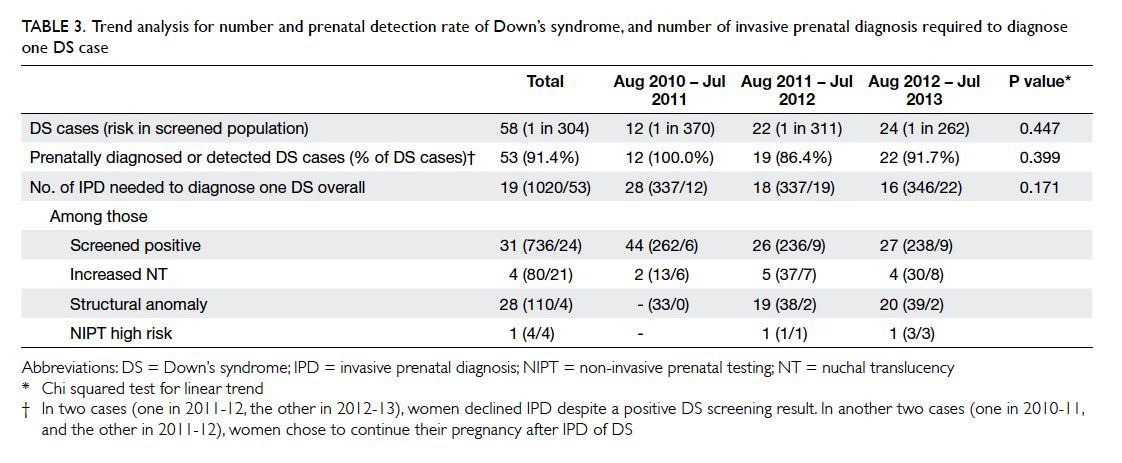
Table 3. Trend analysis for number and prenatal detection rate of Down’s syndrome, and number of invasive prenatal diagnosis required to diagnose one DS case
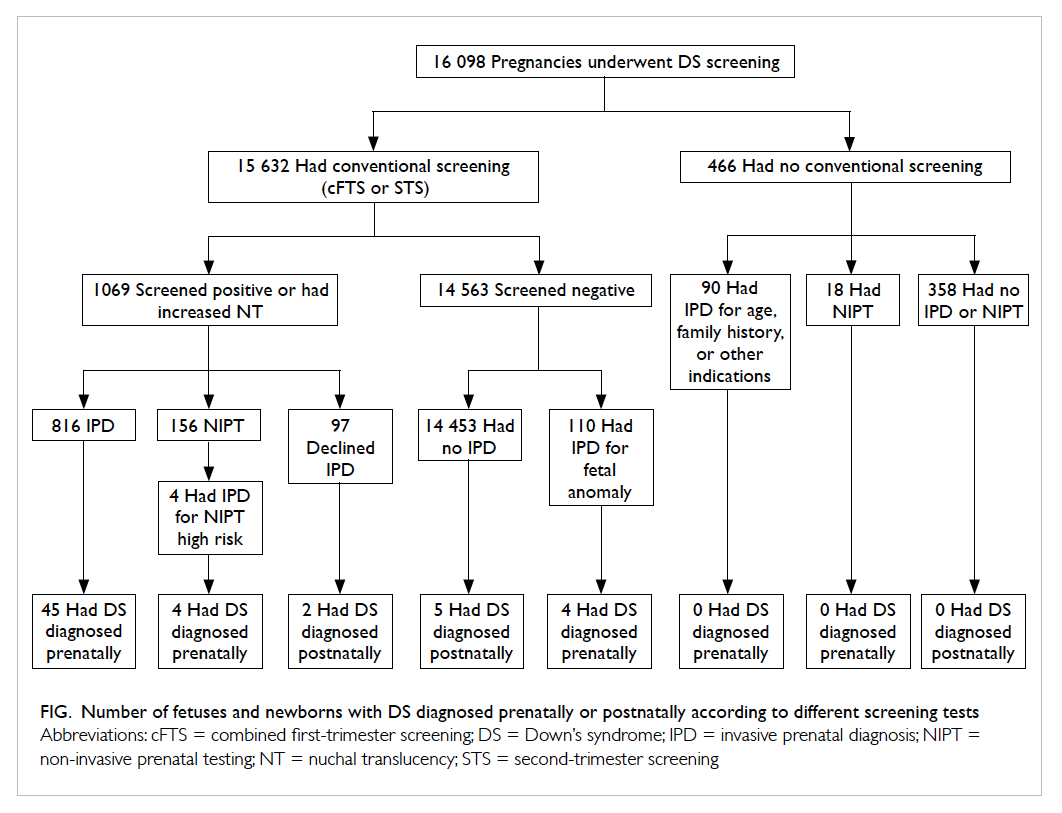
Figure. Number of fetuses and newborns with DS diagnosed prenatally or postnatally according to different screening tests
Performance of non-invasive prenatal testing
Four (2.6%) of 156 women who underwent NIPT for
a screen-positive result (cFTS or STS being 1 in 3, 1 in
25, 1 in 45 and 1 in 230) were considered at high risk
for trisomy 21 (increased amount of chromosome
21 DNA molecules in a maternal sample compared
with that of a euploid reference sample); all results
were confirmed on subsequent IPD. One woman
who had a positive aneuploidy screening but a low-risk
NIPT result underwent IPD and had normal
fetal karyotype. There were no false-negative results
and all babies were confirmed normal after delivery
by routine clinical examination.
Performance of conventional screening
The overall screen-positive and false-positive rates
were 6.8% and 6.5% respectively, and were similar
over the 3-year period (Table 1).
With an increasing number of NIPT as
secondary screening performed for positive cFTS/STS, the false-positive rate of screening decreased
from 6.9% in 2010-11 to 5.2% in 2011-12 and 4.9%
in 2012-13. In 2012-13, with 107 NIPT performed
for a positive cFTS or STS, the false-positive rate
decreased by 29.0% from 6.9% to 4.9%.
The cFTS risk of the five cases of Down’s
syndrome not diagnosed prenatally was 1 in 300,
690, 770, 7300, and 7300. In other words, the
risk of three out of these five cases was below 1 in
1000. All five women were younger than 35 years.
Among those screened negative, four cases of
Down’s syndrome were diagnosed prenatally by
IPD performed for fetal anomaly (Fig). In one of
these four cases, mid-trimester scan showed subtle
sonographic signs including absent nasal bone and
persistent left superior vena cava.
Discussion
As shown in other studies5 14 and our previous study,16
the introduction of NIPT was accompanied by a
decrease in IPD rate. In the present study, we have
further shown that the introduction of NIPT reduced
the IPD rate for positive aneuploidy screening
without affecting the prenatal detection of Down’s
syndrome. Consistent with previous studies,5 13 14
there was a rapid uptake of NIPT, probably because
of its non-invasive nature and high sensitivity and
specificity for common aneuploidies.6 A local study
showed that NIPT results could reduce women’s
uncertainty associated with risk probability–based
results from conventional screening.18 Women are
willing to pay for a test that has a lower false-positive
rate.19
We could not exclude the possibility that the
reduction in IPD rate might be partially related to
an increase in the proportion of cFTS with a lower
false-positive rate than STS.3 Nevertheless, we
observed no significant increase in IPD performed
for structural anomalies despite a concern about
missing atypical chromosomal abnormalities with
NIPT alone.17 20 21
The benefit of reducing the IPD rate is
particularly relevant to our screening programme as
the overall screen-positive rate of our conventional
screening programme was 6.8%, which is higher
than the published figures of 3.3% to 5.9%.4 22 23 24 25 26 With
increasing use of NIPT as secondary screening for
a positive result of cFTS/STS, the false-positive rate
was reduced. The improvement was encouraging
even before full implementation of the strategy using
NIPT as a secondary screening tool.
Assuming 1.8% reduction in IPD (7.6% in
2010-11 – mean of 5.7% in 2011-12 and 6.0%
in 2012-13; Table 1) as in our present study, an
annual delivery rate of 50 000 in Hong Kong,
and 1% miscarriage rate associated with IPD, we
estimate that around 900 IPDs or nine miscarriages
can be potentially avoided if this contingent
approach is adopted widely. This reduction in IPD-related
miscarriage could be further improved as
theoretically about 98% of the IPD for positive
aneuploidy screening could be avoided if NIPT was
used by all screen-positive women.27 Nonetheless,
1.8% ([1020-736]/16 098) of IPD (Table 2) were still
required for other indications including increased
NT or structural anomalies, even if all screen-positive
women opted for NIPT. Alternatively, the
screen-positive rate could be reduced by changing
the cut-off value from 1 in 250 to 1 in 150,2 improving
the quality assurance of measurement of NT (www.fetalmedicine.com) and laboratory assays of serum
markers, algorithms in calculation of trisomy 21
risk, and adding sonographic markers.4 28
The prenatal detection rate of Down’s syndrome
in the present study was similar to the published
results of 83% to 93%.4 22 23 24 25 26 In contrast to cFTS and
STS that have been used in primary screening and
resulted in a reduction in the number of live births
with Down’s syndrome,1 4 introduction of NIPT did not improve the detection rate of our screening
programme. This is expected as NIPT is currently not
routinely used for primary screening. Nevertheless,
NIPT did not decrease the detection rate of Down’s
syndrome as there was no false-negative rate for
NIPT in the present small study. There was concern
about missing atypical abnormalities with NIPT
alone.17 20 21 Further studies are required.
In keeping with international guidelines,29 30 31 32 we suggest offering NIPT as an option to women
with positive aneuploidy screening alone without
increased NT or structural abnormalities to avoid an
unnecessary IPD and its associated miscarriage risk.
We also recommend improving the prenatal detection
rate of a screening programme for Down’s syndrome
by adjusting the cut-off value for cFTS, for example,
from 1 in 250 to 1 in 1000, rather than offering it
to all women as a primary screening.33 In our unit,
the detection rate would be improved from 91.4% to
96.6% as cFTS risk of three of our five missed cases
of Down’s syndrome were above 1 in 1000. As such,
NIPT would be offered to 16.9% of women, including
6.8% with cFTS risk ≥1 in 250 and 10.1% with risk
>1 in 1000 but <1 in 250. Offering an additional
option of NIPT to women with advanced maternal
age only did not improve the detection rate based
on the results of the present study, probably because
all five missed cases were younger than 35 years and
sample size was small. Careful analysis with accurate
assumptions, including the uptake rate of cFTS,
and NIPT, the number of IPD avoided, cut-off value
for cFTS, decreasing charges of NIPT with time,34
and other issues is required to determine the cost-effectiveness
of incorporating NIPT into the current
screening programme for Down’s syndrome.20 35 Major governing or professional bodies recommend
NIPT in the context of informed consent, education,
and pre- and post-test counselling.29 30 31 32 36 In our
previous study,37 we showed that Chinese women
who underwent NIPT recognised the limitations,
but did not understand the complicated aspects.
We suggest giving more information by health care
professionals, preferably trained midwives, so that
patients can make an informed choice.37
The limitations of the present study included
its retrospective nature, single-centre, and small
sample size. The actual performance of NIPT could
not be examined as not all eligible subjects were
tested. Availability and payment methods for NIPT
and other prenatal testing, cut-off level of cFTS, and
women’s preferences differ in different places. Thus, generalisation of the results of the present study
should be done with caution.
Conclusion
The introduction of NIPT as a contingent approach
reduced the IPD rate for positive aneuploidy
screening without increasing the IPD rate for scan
abnormalities or affecting the overall prenatal
detection rate of Down’s syndrome. This fall in IPD
rate was particularly relevant in our centre with a
high false-positive rate after cFTS.
Acknowledgements
We would like to thank the prenatal diagnostic
laboratory of Tsan Yuk Hospital and Prince of
Wales Hospital, Hong Kong for performing the
chromosome analysis.
Declaration
All authors have disclosed no conflicts of interest.
References
1. Cheffins T, Chan A, Haan EA, et al. The impact of maternal
serum screening on the birth prevalence of Down’s
syndrome and the use of amniocentesis and chorionic
villus sampling in South Australia. BJOG 2000;107:1453-9. Crossref
2. Morgan S, Delbarre A, Ward P. Impact of introducing a
national policy for prenatal Down syndrome screening
on the diagnostic invasive procedure rate in England.
Ultrasound Obstet Gynecol 2013;41:526-9. Crossref
3. Muller PR, Cocciolone R, Haan EA, et al. Trends in state/population-based Down syndrome screening and invasive
prenatal testing with the introduction of first-trimester
combined Down syndrome screening, South Australia,
1995-2005. Am J Obstet Gynecol 2007;196:315.e1-7; discussion 285-6.
4. Ekelund CK, Jørgensen FS, Petersen OB, Sundberg K,
Tabor A; Danish Fetal Medicine Research Group. Impact
of a new national screening policy for Down’s syndrome
in Denmark: population based cohort study. BMJ
2008;337:a2547. Crossref
5. Chetty S, Garabedian MJ, Norton ME. Uptake of
noninvasive prenatal testing (NIPT) in women following
positive aneuploidy screening. Prenat Diagn 2013;33:542-6. Crossref
6. Norton ME, Brar H, Weiss J, et al. Non-Invasive
Chromosomal Evaluation (NICE) Study: results of a
multicenter prospective cohort study for detection of
fetal trisomy 21 and trisomy 18. Am J Obstet Gynecol
2012;207:137.e1-8. Crossref
7. Palomaki GE, Kloza EM, Lambert-Messerlian GM, et
al. DNA sequencing of maternal plasma to detect Down
syndrome: an international clinical validation study. Genet
Med 2011;13:913-20. Crossref
8. Bianchi DW, Platt LD, Goldberg JD, Abuhamad AZ, Sehnert
AJ, Rava RP; MatErnal BLood IS Source to Accurately
diagnose fetal aneuploidy (MELISSA) Study Group.
Genome-wide fetal aneuploidy detection by maternal
plasma DNA sequencing. Obstet Gynecol 2012;119:890-901. Crossref
9. Evans MI, Wright DA, Pergament E, Cuckle HS, Nicolaides
KH. Digital PCR for noninvasive detection of aneuploidy:
power analysis equations for feasibility. Fetal Diagn Ther
2012;31:244-7. Crossref
10. Malone FD, Canick JA, Ball RH, et al. First-trimester or
second-trimester screening, or both, for Down’s syndrome.
N Engl J Med 2005;353:2001-11. Crossref
11. Wald NJ. Prenatal screening for open neural tube defects
and Down syndrome: three decades of progress. Prenat
Diagn 2010;30:619-21. Crossref
12. Rozenberg P, Bussières L, Chevret S, et al. Screening for
Down syndrome using first-trimester combined screening
followed by second-trimester ultrasound examination
in an unselected population. Am J Obstet Gynecol
2006;195:1379-87. Crossref
13. Larion S, Romary L, Mlynarczyk M, Abuhamad AZ, Warsof
SL. Changes in prenatal testing trends after introduction
of noninvasive prenatal testing. Obstet Gynecol 2014;123
Suppl 1:62S-63S. Crossref
14. Larion S, Warsof SL, Romary L, Mlynarczyk M, Peleg D,
Abuhamad AZ. Uptake of noninvasive prenatal testing
at a large academic referral center. Am J Obstet Gynecol
2014;211:651.e1-7. Crossref
15. Musci TJ, Fairbrother G, Batey A, Bruursema J, Struble C,
Song K. Non-invasive prenatal testing with cell-free DNA:
US physician attitudes toward implementation in clinical
practice. Prenat Diagn 2013;33:424-8. Crossref
16. Poon CF, Tse WC, Kou KO, Leung KY. Uptake of noninvasive
prenatal testing in Chinese women following
positive Down syndrome screening. Fetal Diagn Ther
2015;37:141-7. Crossref
17. Petersen OB, Vogel I, Ekelund C, Hyett J, Tabor A;
Danish Fetal Medicine Study Group; Danish Clinical
Genetics Study Group. Potential diagnostic consequences
of applying non-invasive prenatal testing: population-based
study from a country with existing first-trimester
screening. Ultrasound Obstet Gynecol 2014;43:265-71. Crossref
18. Yi H, Hallowell N, Griffiths S, Yeung Leung T. Motivations
for undertaking DNA sequencing-based non-invasive
prenatal testing for fetal aneuploidy: a qualitative study
with early adopter patients in Hong Kong. PLoS One
2013;8:e81794. Crossref
19. Lo TK, Lai FK, Leung WC, et al. Screening options for
Down syndrome: how women choose in real clinical
setting. Prenat Diagn 2009;29:852-6. Crossref
20. van Landingham S, Bienstock J, Wood Denne E, Hueppchen
N. Beyond the first trimester screen: can we predict who
will choose invasive testing? Genet Med 2011;13:539-44. Crossref
21. Benn P, Borell A, Chiu R, et al. Position statement from the
Aneuploidy Screening Committee on behalf of the Board
of the International Society for Prenatal Diagnosis. Prenat
Diagn 2013;33:622-9. Crossref
22. Hadlow NC, Hewitt BG, Dickinson JE, Jacoby P, Bower
C. Community-based screening for Down’s Syndrome in
the first trimester using ultrasound and maternal serum
biochemistry. BJOG 2005;112:1561-4. Crossref
23. O’Leary P, Breheny N, Dickinson JE, et al. First-trimester
combined screening for Down syndrome and other fetal
anomalies. Obstet Gynecol 2006;107:869-76. Crossref
24. Soergel P, Pruggmayer M, Schwerdtfeger R, Muhlhaus K,
Scharf A. Screening for trisomy 21 with maternal age, fetal
nuchal translucency and maternal serum biochemistry at
11-14 weeks: a regional experience from Germany. Fetal
Diagn Ther 2006;21:264-8. Crossref
25. Spencer K, Spencer CE, Power M, Dawson C, Nicolaides
KH. Screening for chromosomal abnormalities in the
first trimester using ultrasound and maternal serum
biochemistry in a one-stop clinic: a review of three years
prospective experience. BJOG 2003;110:281-6. Crossref
26. Stenhouse EJ, Crossley JA, Aitken DA, Brogan K, Cameron
AD, Connor JM. First-trimester combined ultrasound and
biochemical screening for Down syndrome in routine
clinical practice. Prenat Diagn 2004;24:774-80. Crossref
27. Chiu RW, Akolekar R, Zheng YW, et al. Non-invasive
prenatal assessment of trisomy 21 by multiplexed maternal
plasma DNA sequencing: large scale validity study. BMJ
2011;342:c7401. Crossref
28. Nicolaides KH. Screening for fetal aneuploidies at 11 to 13
weeks. Prenat Diagn 2011;31:7-15. Crossref
29. American College of Obstetricians and Gynecologists
Committee on Genetics. Committee Opinion No. 545:
Noninvasive prenatal testing for fetal aneuploidy. Obstet
Gynecol 2012;120:1532-4. Crossref
30. Gregg AR, Gross SJ, Best RG, et al. ACMG statement on
noninvasive prenatal screening for fetal aneuploidy. Genet
Med 2013;15:395-8. Crossref
31. Soothill PW, Lo YM. Non-invasive prenatal testing for chromosomal abnormality using maternal plasma DNA, Scientific Impact Paper No. 15. London, Royal College of Obstetricians and Gynaecologists; 2014.
32. Benn P, Borrell A, Chiu R, et al. Position Statement from
the Chromosome Aneuploidy Screening Committee
on behalf of the Board of the International Society for
Prenatal Diagnosis. April 2015. Available from: https://www.ispdhome.org/docs/ISPD/Society%20Statements/PositionStatement_Current_8Apr2015.pdf. Accessed 8 Oct 2015.
33. Benn P, Curnow KJ, Chapman S, Michalopoulos SN,
Hornberger J, Rabinowitz M. An economic analysis of cell-free DNA non-invasive prenatal testing in the US general
pregnancy population. PLoS One 2015;10:e0132313. Crossref
34. Shengmou L, Min C, Chenhong W, et al. Effects, safety
and cost-benefit analysis of Down syndrome screening in
first trimester [in Chinese]. Zhonghua Fu Chan Ke Za Zhi
2014;49:325-30.
35. Stoll K, Lutgendorf M, Knutzen D, Nielsen PE. Questioning
the costs and benefits of non-invasive prenatal testing. J
Matern Fetal Neonatal Med 2014;27:633-4. Crossref
36. Devers PL, Cronister A, Ormond KE, Facio F, Brasington
CK, Flodman P. Noninvasive prenatal testing/noninvasive
prenatal diagnosis: the position of the National Society of
Genetic Counselors. J Genet Couns 2013;22:291-5. Crossref
37. Kou KO, Poon CF, Tse WC, Mak SL, Leung KY. Knowledge
and future preference of Chinese omen in a major public
hospital in Hong Kong after undergoing non-invasive
prenatal testing for positive aneuploidy screening:
a questionnaire survey. BMC Pregnancy Childbirth
2015;15:199. Crossref

